Introduction
Xenograft models are indispensable tools in oncology research, providing a physiologically relevant in vivo platform to study tumor biology, evaluate therapeutic responses, and bridge the translational gap between preclinical experimentation and clinical application. By engrafting human tumor cells or tissues into immunocompromised murine hosts, researchers can recapitulate key aspects of tumor growth, angiogenesis, invasion, and therapeutic resistance within a living system. These models have become essential in developing precision oncology strategies and in shaping regulatory pathways for novel therapeutics.
Historical Perspective and Rationale
The concept of transplanting tumor material across species originated in the early 20th century, but only with the development of immunodeficient mice—such as athymic nude and severe combined immunodeficient (SCID) strains—did xenografts achieve robust and reproducible engraftment. Modern iterations, including NOD scid gamma (NSG) and NOG mice, further improved acceptance of human tissues by eliminating residual innate immune barriers. These breakthroughs provided a stable experimental foundation for preclinical drug development, replacing less predictive two-dimensional in vitro assays.
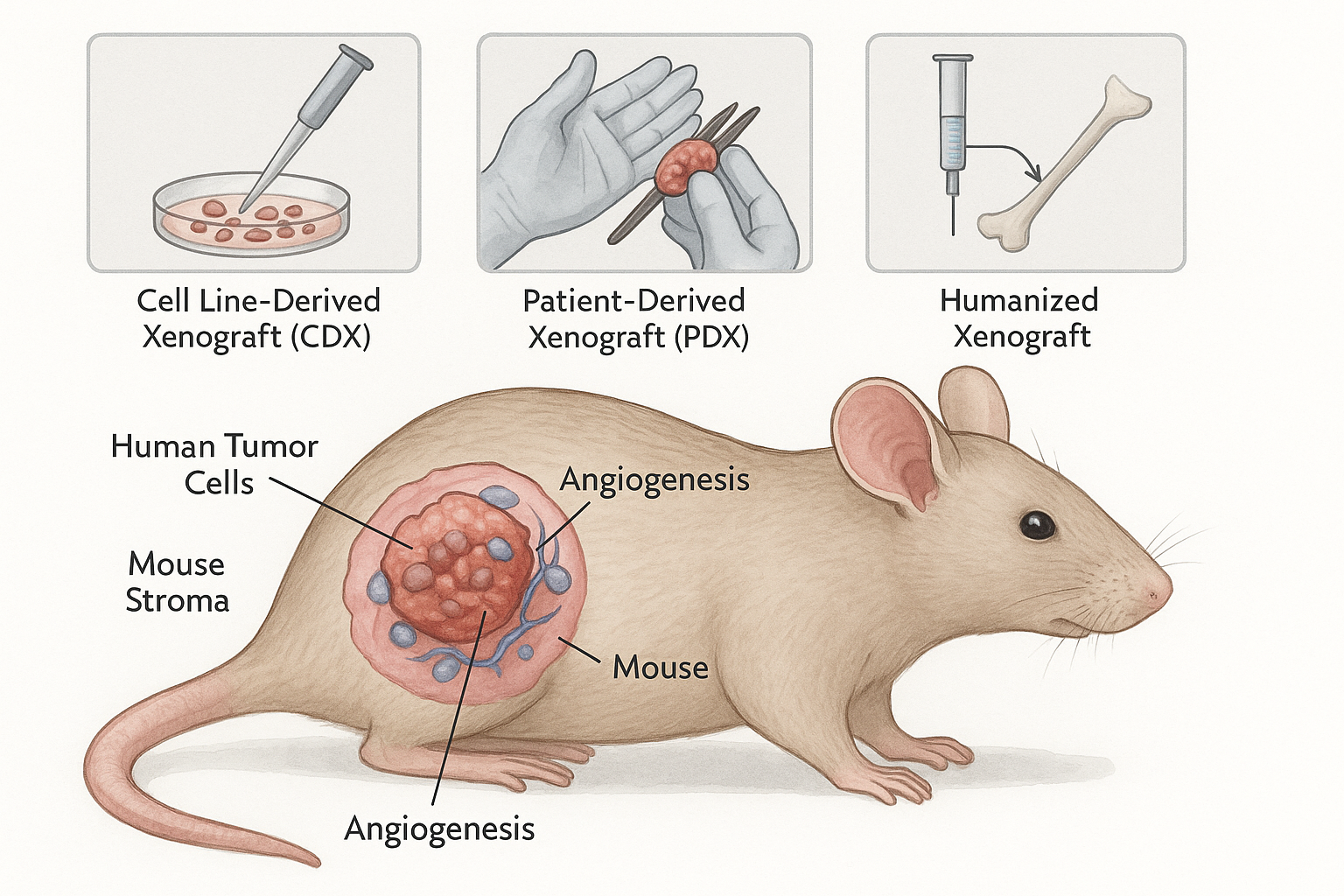
Types of Xenograft Models
- Cell Line–Derived Xenografts (CDX):
Established from immortalized human cancer cell lines, CDX models allow rapid and reproducible tumor growth, high-throughput testing, and ease of handling. However, they often lack the genetic heterogeneity seen in patient tumors.
- Patient-Derived Xenografts (PDX):
Direct implantation of tumor fragments from patients into immunodeficient mice preserves tumor heterogeneity, histological architecture, and molecular features. PDX models serve as a cornerstone of translational oncology, enabling personalized medicine studies and co-clinical trial designs.
- Humanized Xenografts:
By reconstituting murine hosts with human immune systems, these models allow evaluation of immuno-oncology therapeutics, checkpoint inhibitors, and tumor–immune interactions. They represent a crucial advance for immunotherapy development.
Advantages and Limitations
Xenograft models provide unparalleled in vivo relevance for evaluating drug efficacy, resistance, and pharmacokinetics. They allow researchers to explore tumor–stroma interactions, metastatic spread, and the influence of host physiology on tumor biology. However, they also carry limitations: murine stromal replacement can alter tumor microenvironment fidelity, engraftment success is tumor-type dependent, and ethical considerations must be addressed in line with the 3R principles (Replacement, Reduction, Refinement). Additionally, immune-compromised hosts limit the study of tumor–immune dynamics unless specialized humanized systems are employed.
Applications in Preclinical Oncology
- Drug Screening and Mechanism of Action Studies: Xenografts provide a direct readout of therapeutic efficacy and mechanism-driven insights.
- Resistance Modeling: Longitudinal xenograft studies reveal the emergence of resistance pathways, such as activation of bypass signaling or epigenetic reprogramming.
- Biomarker Development: PDX models facilitate identification of predictive biomarkers linked to therapeutic response.
- Co-Clinical Trials: Running PDX studies in parallel with patient trials improves the predictive power of drug response and supports precision oncology strategies.
Future Perspectives
The next generation of xenograft models will increasingly integrate advanced technologies such as CRISPR-based gene editing, single-cell multi-omics, and spatial transcriptomics. Coupled with humanized immune systems and bioengineered scaffolds, these innovations promise to further enhance predictive validity and support the discovery of durable therapeutic interventions. Xenografts will remain a cornerstone of translational oncology as the field continues to move toward individualized and mechanistically precise cancer care.