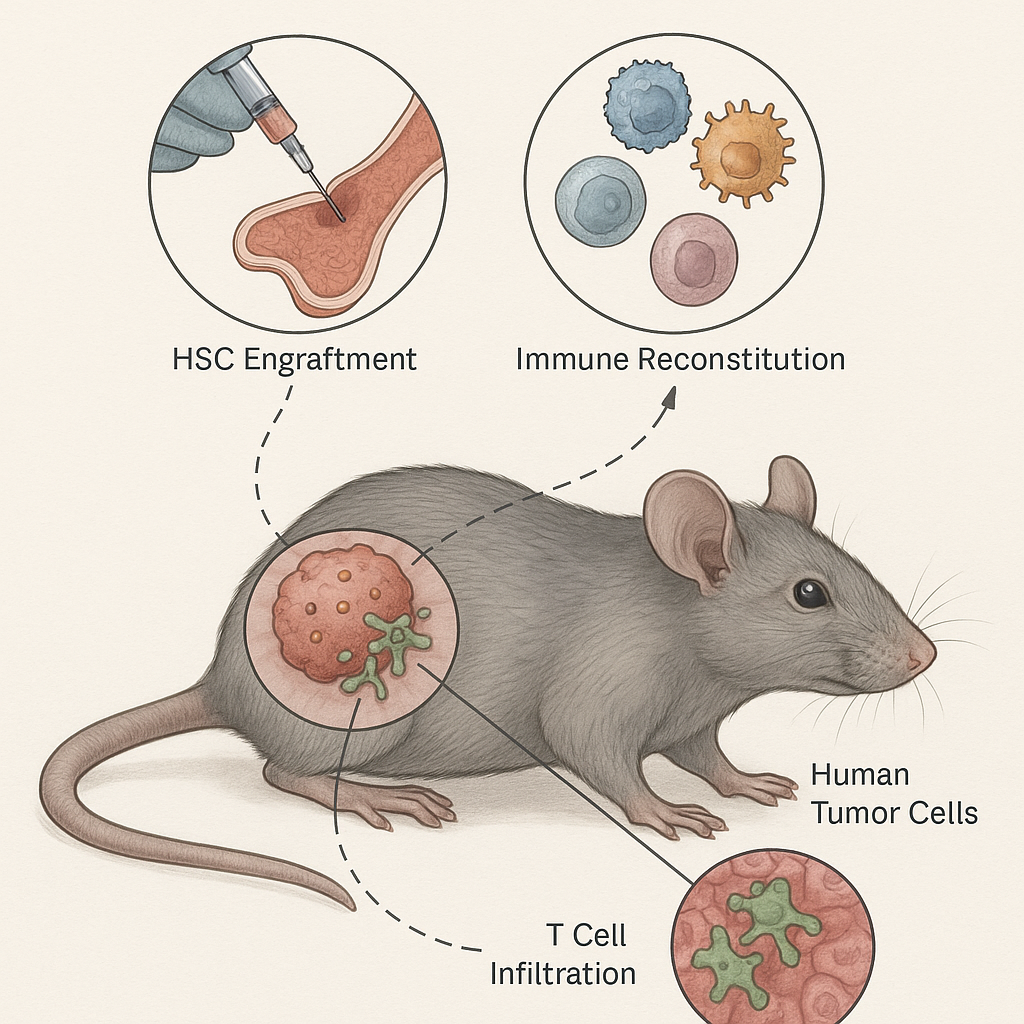
Introduction
Traditional xenograft models provide valuable insights into tumor biology and therapeutic efficacy but are fundamentally limited in their ability to evaluate immunotherapies. Since immunodeficient murine hosts lack functional adaptive immunity, tumor–immune interactions cannot be faithfully studied. Humanized xenograft models overcome this limitation by engrafting mice with components of the human immune system, thereby creating a chimeric platform that supports both human tumor growth and immune response evaluation. These models have become essential for preclinical testing of checkpoint inhibitors, adoptive cell therapies, and cancer vaccines.
Approaches to Humanization
Two major strategies dominate the generation of humanized xenografts:
- Hematopoietic Stem Cell (HSC)-Based Humanization:
- NSG or NOG mice are irradiated and transplanted with CD34⁺ human hematopoietic stem cells (often derived from cord blood).
- Over time, these cells reconstitute multilineage human immune compartments, including T cells, B cells, NK cells, and antigen-presenting cells.
- Peripheral Blood Mononuclear Cell (PBMC)-Based Humanization:
- Engraftment with human PBMCs provides rapid immune reconstitution but is often associated with graft-versus-host disease (GVHD).
- This approach is useful for short-term immunotherapy efficacy studies.
Key Applications
- Checkpoint Inhibitors: Evaluating anti-PD-1, anti-PD-L1, and anti-CTLA-4 agents in a human immune context.
- Cell-Based Therapies: Testing chimeric antigen receptor (CAR) T-cell function and persistence in vivo.
- Cancer Vaccines: Studying antigen-specific T-cell priming and immune memory formation.
- Cytokine Therapies: Assessing activity of IL-2, IL-12, or GM-CSF in modulating tumor–immune interactions.
Advantages
- Recapitulate human tumor–immune cell interactions.
- Enable identification of immune-related biomarkers of therapeutic response.
- Support co-clinical testing of next-generation immunotherapies.
Limitations
- Partial immune system reconstitution, often skewed toward lymphoid over myeloid compartments.
- Risk of GVHD in PBMC-based models.
- Engraftment efficiency varies by donor source and host strain.
- Cost- and labor-intensive procedures requiring specialized facilities.
Future Perspectives
Emerging technologies are enhancing the predictive fidelity of humanized xenografts. Approaches such as CRISPR-mediated editing of murine cytokine genes, transgenic expression of human HLA alleles, and microbiome modulation are being integrated to improve immune functionality. In the coming years, humanized xenograft systems are expected to become indispensable platforms for preclinical immuno-oncology, accelerating the development of durable, patient-tailored immune therapies.