Toledo Xenograft Model Overview
The Toledo xenograft model is established from a human non-Hodgkin lymphoma (NHL) cell line classified under diffuse large B-cell lymphoma (DLBCL) of the germinal center B-cell–like (GCB) subtype. Originating from a relapsed NHL patient, the Toledo cell line exhibits classic features of mature B lymphocytes and demonstrates sensitivity to immunotherapy and chemotherapeutic regimens used in clinical treatment of GCB-DLBCL. The xenograft model recapitulates both the morphological and molecular landscape of its clinical counterpart and is widely employed for preclinical validation of monoclonal antibodies, targeted agents, and emerging combination therapies for aggressive lymphomas.
Request a Custom Quote for Toledo Xenograft ModelBiological and Molecular Characteristics
Toledo cells display a prototypical GCB-DLBCL immunophenotype, including expression of surface markers CD19, CD20, CD10, and BCL6, with absence of MUM1. The cells are EBV-negative and retain surface immunoglobulin, making them suitable for studying B-cell receptor–dependent signaling and associated vulnerabilities. High levels of BCL2 and MHC class II molecules are expressed, supporting investigations into anti-apoptotic signaling and immune-mediated cytotoxicity. The Toledo cell line also demonstrates MYC expression, contributing to enhanced proliferation and resistance patterns. Its defined GCB lineage and stable molecular features enable subtype-specific therapeutic testing and biomarker discovery.
| Characteristic | Description |
|---|---|
| Tissue Origin | Human diffuse large B-cell lymphoma (GCB subtype) |
| Immunophenotype | CD19+, CD20+, CD10+, BCL6+, MUM1–, HLA-DR+, sIg+ |
| Viral Status | EBV-negative |
| Key Molecular Features | BCL2+, MYC+, GCB signature |
| Clinical Relevance | CD20-targeted therapy, BCL2 inhibitors, chemoimmunotherapy |
In Vivo Model Development and Tumorigenicity
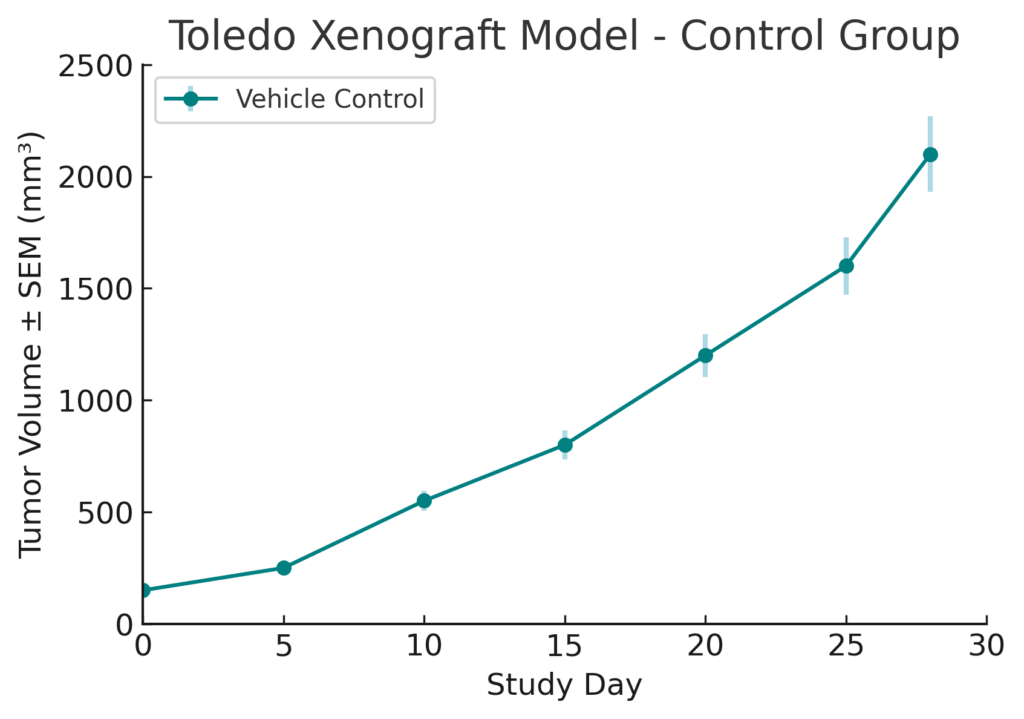
The Toledo xenograft model is developed by subcutaneous injection of cultured cells into immunodeficient mouse strains such as NOD/SCID or NSG. Tumor formation is robust, with visible nodules emerging by 2 to 3 weeks post-inoculation and reaching volumes between 500–900 mm³ over 6 to 8 weeks. This model exhibits consistent tumor take rates and growth kinetics, permitting reproducible assessment of investigational agents. The model’s predictability and molecular alignment with clinical GCB-DLBCL make it an optimal system for high-throughput screening and longitudinal therapeutic monitoring.
Request a Custom Quote for Toledo Xenograft ModelHistopathology and Immunohistochemical Profile
Toledo xenografts are composed of medium-to-large lymphoid cells with round nuclei, dispersed chromatin, and frequent mitotic figures. Hematoxylin and eosin staining reveals a diffuse architecture lacking follicular structures. Immunohistochemical staining demonstrates strong membranous CD20 and nuclear BCL6 expression, with high Ki-67 labeling (typically >70%) reflecting aggressive proliferative behavior. BCL2 and MHC class II positivity are consistently observed, underscoring the model’s utility for both apoptosis modulation studies and immune recognition assays. The histologic features remain stable across generations and support translational fidelity to patient-derived tumors.
Preclinical Applications and Drug Response
The Toledo model is widely used in preclinical studies targeting GCB-DLBCL with monoclonal antibodies (e.g., rituximab, obinutuzumab), BCL2 antagonists (e.g., venetoclax), and PI3K/AKT pathway inhibitors. Its predictable response to CD20-directed therapies, combined with expression of MYC and BCL2, makes it suitable for evaluating rational drug combinations aimed at overcoming resistance and improving treatment durability. This model also provides a platform for immunotherapeutic strategies, including bispecific antibodies and checkpoint blockade, due to preserved antigen presentation and immune-relevant markers. Overall, the Toledo xenograft is a robust and translationally relevant model for studying GCB-DLBCL pathophysiology and therapeutic response.
Request This Model
To request the Toledo xenograft model for your preclinical studies, please use the form below. A customized quote and additional model specifications will be provided upon inquiry.
Request a Custom Quote for Toledo Xenograft Model