T.Tn Xenograft Model Overview
The T.Tn xenograft model is derived from a human colorectal adenocarcinoma cell line originally isolated from a primary tumor in a 70-year-old male patient. Recognized for its aggressive growth and moderately differentiated epithelial phenotype, the T.Tn model is particularly valuable in preclinical studies aimed at understanding colorectal cancer progression, therapeutic resistance, and drug efficacy in microsatellite-stable (MSS) tumors. The model’s stable engraftment, reliable tumor growth kinetics, and translational relevance to standard colorectal cancer pathophysiology make it a suitable platform for evaluating monotherapies and combination regimens, especially those targeting EGFR signaling, apoptosis regulation, and downstream oncogenic pathways.
Request a Custom Quote for T.Tn Xenograft ModelBiological and Molecular Characteristics
T.Tn cells exhibit cohesive, epithelial morphology and retain classical markers of colorectal origin. The cell line is microsatellite stable (MSS) and carries a KRAS mutation—commonly reported as G12D—which renders it intrinsically resistant to EGFR inhibitors such as cetuximab and panitumumab. TP53 is also mutated in this model, impairing apoptotic responses and allowing for the study of DNA damage tolerance and synthetic lethality. Expression of carcinoembryonic antigen (CEA) and cytokeratin 20 (CK20) is detectable, supporting the cell line’s colorectal lineage. Wnt/β-catenin signaling is active, with partial nuclear localization of β-catenin observed in both in vitro cultures and xenograft tissues. These molecular attributes make the T.Tn model particularly useful for testing drugs targeting RAS/MAPK and PI3K/AKT signaling pathways, as well as agents modulating apoptotic resistance.
| Characteristic | T.Tn Cell Line Profile |
|---|---|
| Tissue of Origin | Colorectal adenocarcinoma (primary) |
| KRAS Status | Mutant (G12D) |
| TP53 Status | Mutated |
| MSI Status | Microsatellite stable (MSS) |
| Differentiation Markers | CK20, CEA, E-cadherin |
| Wnt Signaling | Active, β-catenin cytoplasmic/nuclear |
In Vivo Model Development and Tumorigenicity
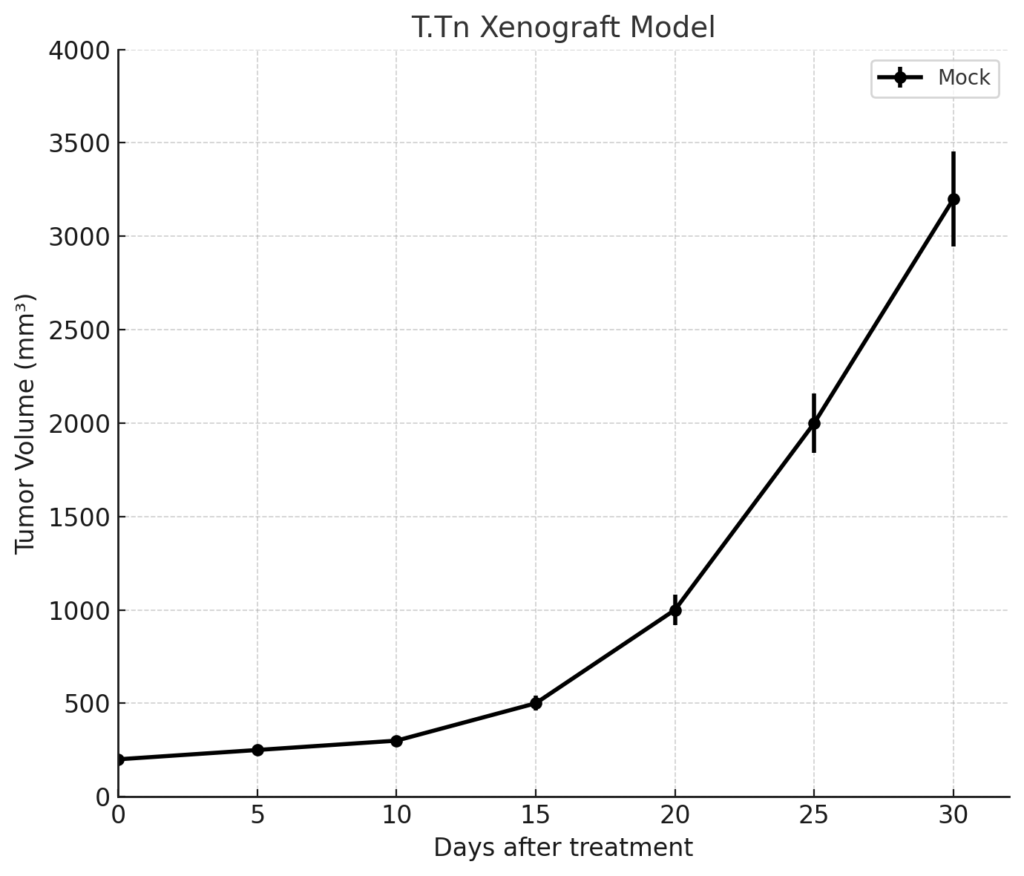
T.Tn xenografts are typically established by subcutaneous implantation of cultured cells into immunodeficient mouse strains, including athymic nude or NOD/SCID models. Tumor growth is initiated within 8 to 12 days post-injection, and tumors consistently reach target volumes of 700–900 mm³ within 4 to 5 weeks. The model demonstrates a moderate to high growth rate and supports extended treatment studies evaluating drug efficacy and resistance development. Due to the presence of a KRAS mutation, T.Tn xenografts are often employed in experiments designed to bypass or overcome EGFR resistance through the use of MEK, ERK, or PI3K inhibitors. The model’s consistent tumor architecture and defined molecular profile make it suitable for studies requiring repeatable, genotype-specific endpoints.
Request a Custom Quote for T.Tn Xenograft ModelHistopathology and Immunohistochemical Profile
Histologically, T.Tn xenograft tumors present as moderately differentiated colorectal adenocarcinomas with glandular structures, luminal necrosis, and defined epithelial polarity. Hematoxylin and eosin (H&E) staining reveals columnar tumor cells with apical cytoplasm and basal nuclei, mirroring the organization of intestinal epithelial tissue. Immunohistochemical staining confirms strong membrane-localized E-cadherin, moderate cytoplasmic β-catenin, and focal nuclear accumulation, indicating active Wnt pathway signaling. Tumors stain positively for CK20 and CEA, confirming colorectal lineage. p53 accumulation is frequently observed in nuclei, consistent with TP53 mutation. The low stromal content and lack of extensive necrosis contribute to uniformity across samples, making this model highly amenable to histopathologic and biomarker evaluation.
Preclinical Applications and Drug Response
The T.Tn xenograft model is widely applied in studies of KRAS-mutant colorectal cancer, where resistance to EGFR-targeted therapies is a key challenge. It serves as a valuable platform for testing small molecule inhibitors of downstream pathways, including MEK, ERK, and mTOR, and is often used in combination therapy studies designed to overcome RAS-driven oncogenesis. Due to its TP53 mutation, the model also supports the evaluation of DNA-damaging agents, as well as novel compounds that exploit defects in the apoptotic response. T.Tn xenografts are further utilized in biomarker discovery efforts, drug synergy investigations, and preclinical validation of targeted therapies aimed at non-EGFR-sensitive CRC populations. Its consistent tumorigenic profile, defined molecular landscape, and relevance to resistant colorectal subtypes make it a reliable system for translational oncology research.
Request This Model
To incorporate the T.Tn xenograft model into your colorectal cancer research or drug development pipeline, contact our scientific team to request detailed model specifications and initiate a custom study design tailored to your experimental objectives.
Request a Custom Quote for T.Tn Xenograft Model