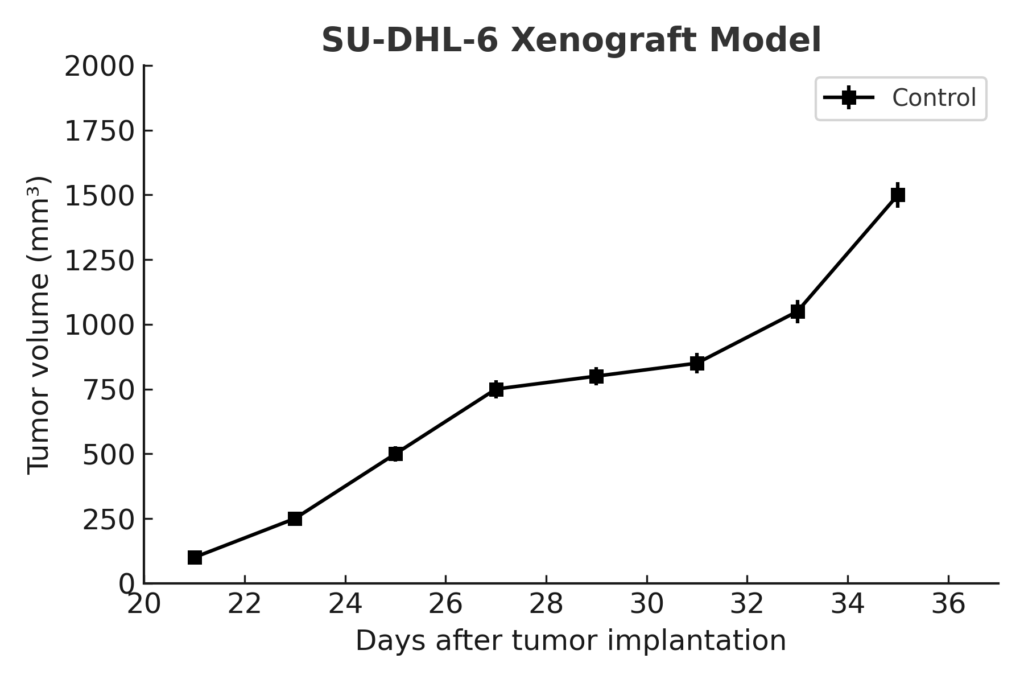
SU‑DHL‑6 Xenograft Model Overview
SU‑DHL‑6 is a germinal center B‑cell–like (GCB) subtype of diffuse large B‑cell lymphoma (DLBCL), widely used in translational oncology as an in vivo model of BCL2-driven, apoptosis-resistant lymphoma. Originally derived from a large-cell lymphoma patient, this cell line harbors the signature t(14;18) translocation and demonstrates BCL2 overexpression, consistent with its germinal center origin. When implanted subcutaneously into immunocompromised mice, SU‑DHL‑6 reliably forms tumors and supports robust preclinical studies evaluating targeted therapeutics, epigenetic regulators, and immune-enabled combination therapies.
Request a Custom Quote for SU‑DHL‑6 Xenograft ModelBiological and Molecular Characteristics
SU‑DHL‑6 carries the t(14;18)(q32;q21) translocation, resulting in constitutive BCL2 expression and resistance to apoptosis. It lacks mutations in MYD88 or CD79B, features common to ABC‑DLBCL, but exhibits wild‑type TP53. CREBBP mutations are often present, which confer susceptibility to chromatin remodeling therapies. The cell line expresses CD19, CD20, CD10, and retains both MHC I and II expression, supporting studies in immuno‑oncology settings. Its baseline PD‑L1 expression is low, making this model suitable for immune‑priming and monoclonal antibody evaluation (cellosaurus.org).
| Characteristic | Description |
|---|---|
| Tissue Origin | Human germinal center B‑cell–like DLBCL |
| Key Alterations | BCL2 t(14;18) translocation; CREBBP mutation |
| Mutation Status | TP53 wild‑type; MYD88/CD79B wild‑type |
| Immunophenotype | CD19+, CD20+, CD10+, MHC I/II+, low PD‑L1 |
| Therapeutic Relevance | BCL2 inhibition, epigenetic therapies, CD20-targeted agents |
In Vivo Model Development and Tumorigenicity
SU‑DHL‑6 tumors are established by subcutaneous injection into NOD/SCID or NSG mice, typically requiring 2–3 weeks for tumor formation. Once palpable, tumors grow consistently to 400–600 mm³ over 5–7 weeks, providing reproducible conditions for drug efficacy and biomarker studies. The model’s moderate growth rate and defined genetic profile make it ideal for evaluating single‑agent and combination regimens targeting apoptotic pathways, epigenetic modifiers, and monoclonal antibodies.
Request a Custom Quote for SU‑DHL‑6 Xenograft ModelHistopathology and Immunohistochemical Profile
Histologic evaluation of SU‑DHL‑6 xenografts reveals sheets of large atypical lymphoid cells with vesicular nuclei, prominent nucleoli, and frequent mitoses. Immunohistochemistry shows strong CD20 and BCL6 expression, diffuse cytoplasmic BCL2, and nuclear BCL6 as well as downregulated acetylated histone H3 in cell lines with CREBBP mutation. Ki‑67 labeling indexes range from 60–70%, consistent with moderate-to-high proliferation. MHC class II expression is preserved, supporting translational modeling of antigen presentation and immunotherapy strategies.
Preclinical Applications and Drug Response
SU‑DHL‑6 has been extensively employed in preclinical testing of venetoclax and other BCL2 inhibitors, often in combination with epigenetic modulators such as EZH2 or HDAC inhibitors, especially in the presence of CREBBP mutation. BET inhibitors like JQ1 have demonstrated efficacy in suppressing MYC-driven proliferation even in GCB DLBCL lines including SU‑DHL‑6 (pmc.ncbi.nlm.nih.gov). The model is also used to evaluate antibody-based therapies (e.g., anti-CD20), bispecific constructs, and immune sensitization strategies leveraging preserved antigen presentation and low PD-L1 expression. Studies combining CXCR4 inhibitors and BET therapies have shown synergy across both GCB and ABC subtypes, including SU‑DHL‑6 (haematologica.org), underlining the model’s versatility in testing rational combinatorial interventions.
Request This Model
To request the SU‑DHL‑6 xenograft model for your preclinical studies, please use the form below. A customized quote and additional model specifications will be provided upon inquiry.
Request a Custom Quote for SU‑DHL‑6 Xenograft Model