SU-DHL-4 Xenograft Model Overview
The SU-DHL-4 xenograft model is derived from a human diffuse large B-cell lymphoma (DLBCL) of the germinal center B-cell–like (GCB) subtype and is widely used to evaluate preclinical responses to targeted agents, epigenetic therapies, and monoclonal antibodies. Originating from a male patient, the SU-DHL-4 cell line displays molecular features characteristic of GCB-DLBCL, including BCL2 translocation and TP53 mutations. Unlike ABC-subtype DLBCLs, SU-DHL-4 is less dependent on chronic active B-cell receptor signaling and instead demonstrates sensitivity to BCL2 inhibition and chromatin remodeling agents. When implanted subcutaneously in immunodeficient mice, SU-DHL-4 generates highly reproducible tumor growth, supporting therapeutic evaluation in a genomically defined lymphoma context.
Request a Custom Quote for SU‑DHL‑4 Xenograft ModelBiological and Molecular Characteristics
The SU-DHL-4 cell line retains key genetic and immunophenotypic features of GCB-DLBCL. Most notably, it harbors the t(14;18)(q32;q21) translocation resulting in BCL2 overexpression, contributing to resistance against apoptosis. TP53 is mutated, and the cells do not carry MYD88 or CD79B mutations, distinguishing this model from ABC-DLBCL counterparts. The immunophenotype includes strong expression of CD19, CD20, and CD10, consistent with a germinal center origin. These cells also express MHC class I and II molecules, supporting use in immunological and antigen-presentation studies. PD-L1 expression is low, which enhances the model’s utility for immune-sensitization or checkpoint blockade combinations.
| Characteristic | Description |
|---|---|
| Tissue Origin | Human germinal center B-cell–like DLBCL |
| Key Alterations | BCL2 translocation, TP53 mutation |
| Mutation Status | MYD88 and CD79B wild-type |
| Immunophenotype | CD19+, CD20+, CD10+, MHC I/II+, PD-L1 (low) |
| Therapeutic Relevance | GCB-DLBCL biology, epigenetic and BCL2-targeted therapies |
In Vivo Model Development and Tumorigenicity
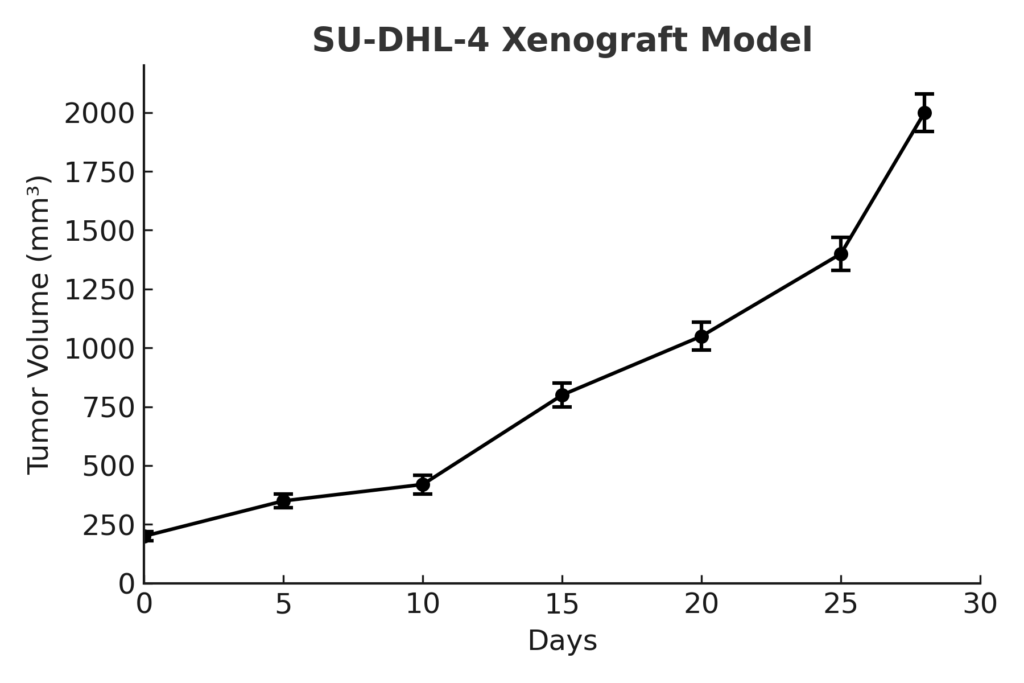
The SU-DHL-4 xenograft model is established by subcutaneous injection of tumor cells into immunodeficient mouse strains such as NSG or NOD/SCID mice. Tumors typically become visible within 2–3 weeks post-inoculation and expand steadily, reaching volumes of 400–600 mm³ over a 5–7 week period. The model exhibits high tumor take rates and predictable growth curves, making it suitable for comparative therapeutic studies. SU-DHL-4 is particularly effective for testing the efficacy of BCL2 inhibitors like venetoclax, as well as HDAC inhibitors, EZH2 antagonists, and combinations with anti-CD20 monoclonal antibodies.
Request a Custom Quote for SU‑DHL‑4 Xenograft ModelHistopathology and Immunohistochemical Profile
Histologically, SU-DHL-4 xenografts exhibit large, atypical lymphoid cells arranged in diffuse sheets with vesicular nuclei and prominent nucleoli. Hematoxylin and eosin staining highlights dense tumor architecture with occasional necrosis and minimal stromal involvement. Immunohistochemical profiling shows strong and diffuse CD20 expression, high nuclear BCL2 staining due to t(14;18) translocation, and nuclear positivity for BCL6, consistent with GCB origin. Ki-67 staining reveals a proliferation index of approximately 60–70%, indicative of moderate to high mitotic activity. MHC class II expression is preserved, providing a platform for immuno-oncology approaches focused on enhancing tumor immunogenicity.
Preclinical Applications and Drug Response
The SU-DHL-4 xenograft model is extensively used to evaluate BCL2-targeted therapies, epigenetic modulators, and antibody-based treatments for GCB-DLBCL. The presence of the BCL2 translocation makes the model particularly responsive to BH3 mimetics like venetoclax. It has also demonstrated sensitivity to EZH2 inhibitors, especially in the context of chromatin remodeling studies. The model supports combination regimens that include anti-CD20 monoclonal antibodies (e.g., rituximab) and histone deacetylase (HDAC) inhibitors. Due to its relatively intact antigen presentation machinery and low PD-L1 expression, it is also suitable for immune-enhancing strategies, including T-cell engagers and immune checkpoint adjuvants.
Request This Model
To request the SU-DHL-4 xenograft model for your preclinical studies, please use the form below. A customized quote and additional model specifications will be provided upon inquiry.
Request a Custom Quote for SU‑DHL‑4 Xenograft Model