SU-DHL-1 Xenograft Model Overview
The SU-DHL-1 xenograft model is derived from a human anaplastic large cell lymphoma (ALCL) cell line, established from the pleural effusion of a patient with diffuse large-cell lymphoma exhibiting T/null-cell characteristics. This model is notable for its expression of the oncogenic fusion protein NPM1-ALK, a defining molecular hallmark of ALK-positive ALCL. The SU-DHL-1 xenograft is an established platform for studying lymphoid malignancies with ALK translocations and serves as a robust in vivo system for preclinical evaluation of ALK inhibitors, immunotherapies, and cell cycle regulators in aggressive T-cell neoplasms. Its strong tumorigenicity, genetic stability, and high proliferative rate enable consistent and translationally relevant experimental outcomes.
Request a Custom Quote for SU‑DHL‑1 Xenograft ModelBiological and Molecular Characteristics
SU-DHL-1 cells represent a T/null-cell lymphoma phenotype, exhibiting CD30 expression and ALK gene rearrangement resulting in the NPM1-ALK fusion transcript. This fusion leads to constitutive activation of ALK tyrosine kinase, which drives downstream signaling through pathways including STAT3, PI3K/AKT, and MAPK, promoting uncontrolled proliferation and resistance to apoptosis. The immunophenotypic profile includes CD30+, CD4+, CD2+, CD45+, and ALK+, while lacking surface T-cell receptor expression. The model also expresses high levels of anti-apoptotic proteins such as BCL2 and survivin, contributing to therapeutic resistance. Its reproducible molecular characteristics make SU-DHL-1 ideal for mechanism-based drug testing.
| Characteristic | Description |
|---|---|
| Disease Origin | Human anaplastic large cell lymphoma (ALK-positive) |
| Genetic Alteration | NPM1-ALK fusion via t(2;5)(p23;q35) translocation |
| Immunophenotype | CD30+, ALK+, CD45+, CD2+, CD4+, TCR– |
| Key Signaling Pathways | ALK, STAT3, PI3K/AKT, MAPK |
| Therapeutic Relevance | ALK inhibitors, CD30-directed therapies, apoptosis modulators |
In Vivo Model Development and Tumorigenicity
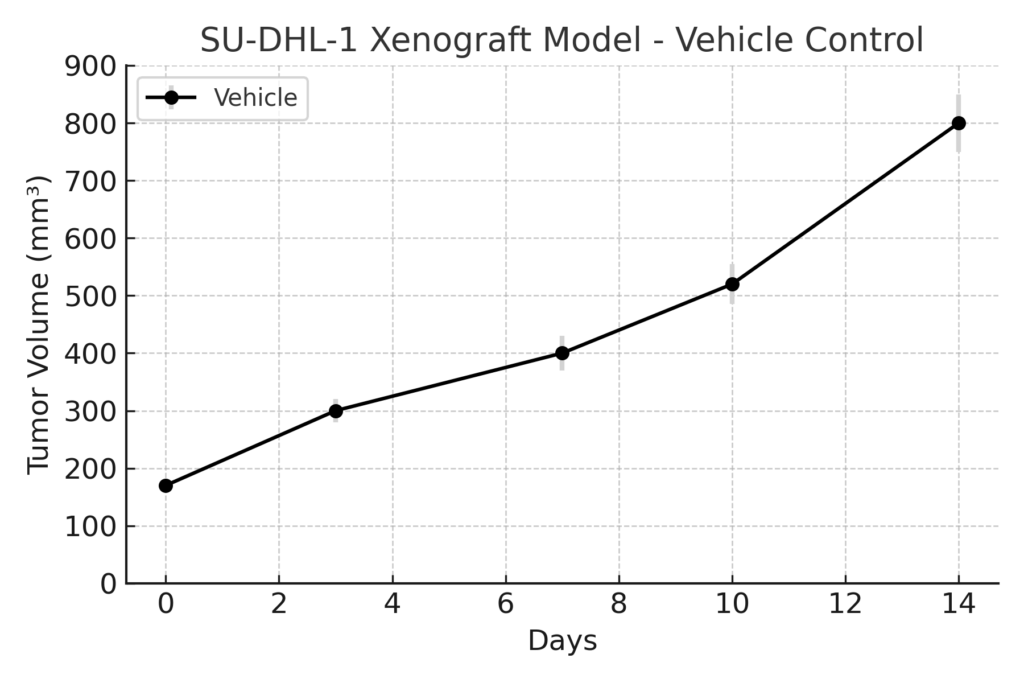
The SU-DHL-1 xenograft is typically established through subcutaneous injection of cultured cells into immunodeficient mice such as NOD/SCID or NSG. Tumor engraftment is rapid, with nodules forming within two weeks and reaching measurable volumes between 500–900 mm³ within six weeks. The model demonstrates near-complete tumor take and uniform growth kinetics, enabling consistent cohort-based analyses. Its reproducibility and ALK-driven biology provide a controlled environment for assessing dose-response, pharmacokinetics, and molecular biomarker modulation during drug treatment studies targeting T-cell lymphomas.
Request a Custom Quote for SU‑DHL‑1 Xenograft ModelHistopathology and Immunohistochemical Profile
Histologically, SU-DHL-1 xenografts consist of large pleomorphic lymphoid cells with abundant cytoplasm, horseshoe- or kidney-shaped nuclei, and prominent nucleoli—morphological features consistent with ALCL. Hematoxylin and eosin staining reveals cohesive sheets of tumor cells without follicular or nodal architecture. Immunohistochemical analysis shows strong membranous and cytoplasmic CD30 staining, nuclear and cytoplasmic ALK positivity, and robust expression of CD45. A high Ki-67 index (>80%) is observed, indicating aggressive proliferative behavior. These findings closely recapitulate the histologic features of ALK-positive ALCL, supporting their utility in translational preclinical investigations.
Preclinical Applications and Drug Response
The SU-DHL-1 xenograft model has been widely used in preclinical studies of ALK inhibitors (e.g., crizotinib, ceritinib, brigatinib) and anti-CD30 monoclonal antibody-drug conjugates such as brentuximab vedotin. It is also employed in exploring STAT3 inhibitors, proteasome inhibitors, and HDAC inhibitors due to its dysregulated epigenetic and signaling landscapes. Combination therapy studies involving ALK blockade and BCL2 inhibition are supported by the model’s high BCL2 expression. Moreover, the model is used to evaluate resistance mechanisms to ALK-targeted agents and to test novel approaches in T-cell lymphoma immunotherapy.
Request This Model
To request the SU‑DHL‑1 xenograft model for your preclinical studies, please use the form below. A customized quote and additional model specifications will be provided upon inquiry.
Request a Custom Quote for SU‑DHL‑1 Xenograft Model