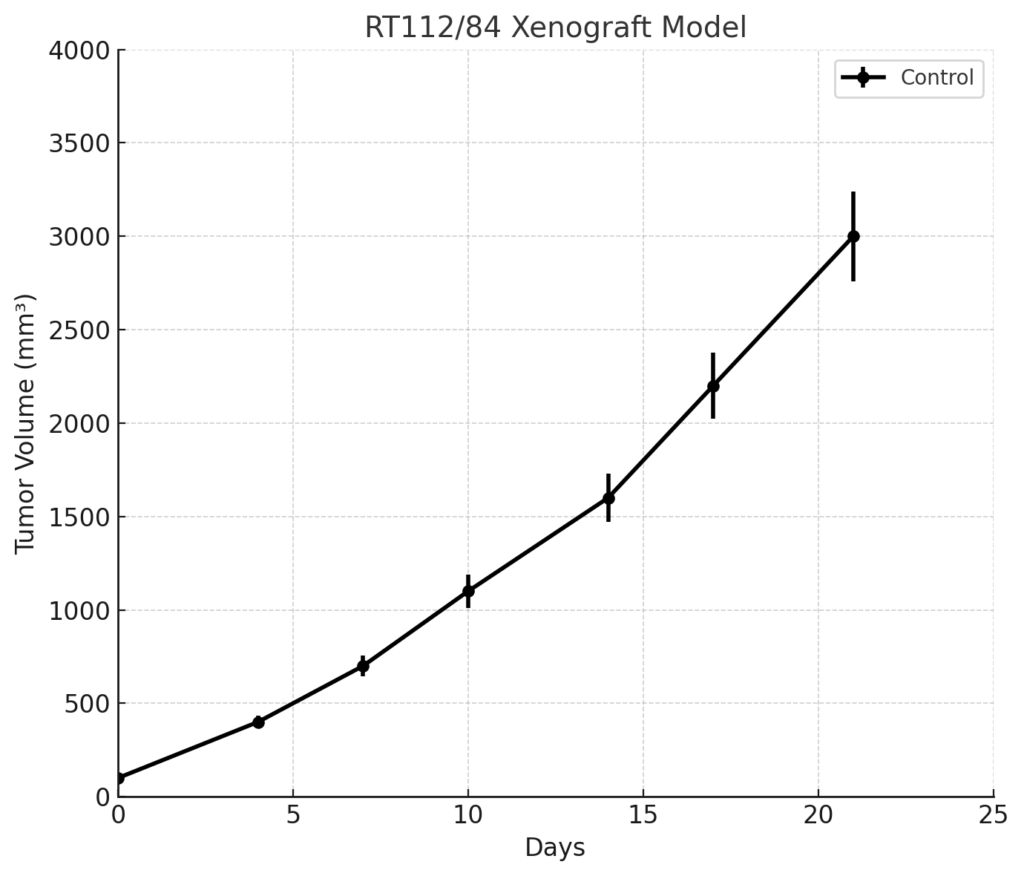
RT112/84 Xenograft Model Overview
The RT112/84 xenograft model is derived from the RT112 human bladder carcinoma cell line, originally established from a patient with grade II transitional cell carcinoma. As a moderately differentiated, luminal-type urothelial cancer model, RT112/84 retains key features of its parental lineage, including epithelial morphology, expression of differentiation-associated markers, and a relatively indolent growth profile in vivo. The RT112 cell line is well recognized for its suitability in modeling early-stage or non–muscle-invasive bladder carcinoma (NMIBC), providing a sharp contrast to more aggressive models such as T24 or UM-UC-3.
The RT112/84 variant used in xenograft studies offers improved consistency in tumor take rate, stable in vivo growth kinetics, and robust epithelial differentiation, making it particularly valuable for investigations into receptor tyrosine kinase signaling, FGFR3-targeted therapies, and urothelial differentiation dynamics. This model provides a unique opportunity to assess drug responses in a biologically distinct subtype of bladder cancer, especially relevant to therapeutic strategies aimed at luminal tumors exhibiting FGFR3 alterations and E-cadherin retention.
Request a Custom Quote for RT112/84 Xenograft ModelBiological and Molecular Characteristics
RT112/84 cells are characterized by a relatively stable genome, with amplification of the FGFR3 gene and high levels of FGFR3 protein expression, a molecular alteration frequently observed in low- to intermediate-grade bladder tumors. These cells maintain expression of epithelial markers such as E-cadherin, cytokeratin 20, and uroplakin II, indicating a differentiated phenotype. Unlike high-grade invasive lines, RT112/84 does not harbor mutations in TP53 or HRAS, and demonstrates functional p53 activity, which correlates with intact apoptotic and DNA damage responses.
Expression of HER2/neu (ERBB2) and EGFR is detectable at moderate levels, though downstream signaling activity remains less aggressive compared to invasive models. RT112/84 cells also retain Rb function and show regulated cell cycle transitions. The luminal differentiation status is supported by a high expression of GATA3 and FOXA1, transcription factors associated with bladder epithelial lineage specification.
The table below summarizes key biological features of the RT112/84 cell line:
| Characteristic | RT112/84 Profile |
|---|---|
| Origin | Human urinary bladder, grade II transitional carcinoma |
| FGFR3 Status | Amplified and overexpressed |
| TP53 Status | Wild-type, functionally intact |
| pRb Status | Wild-type |
| HER2/EGFR Expression | Moderate |
| Differentiation Markers | E-cadherin+, CK20+, uroplakin II+ |
| Luminal Markers | High GATA3, FOXA1 |
| EMT Profile | Negative; no mesenchymal transition |
| Cytogenetics | Near-diploid; low chromosomal instability |
| Invasive Potential (in vitro) | Low |
This molecular configuration positions RT112/84 as an ideal model for evaluating therapeutic candidates targeting FGFR3-driven or luminal-differentiated urothelial tumors, as well as for elucidating early oncogenic events in urothelial transformation.
In Vivo Model Development and Tumorigenicity
The RT112/84 xenograft model displays reproducible subcutaneous tumor formation in immunodeficient mice, including athymic nude and NOD-SCID strains. Tumor engraftment typically occurs within 14–18 days following implantation of 5 × 10^6 to 1 × 10^7 cells in a matrix-supporting medium such as Matrigel. Compared to more aggressive bladder cancer models, RT112/84 demonstrates slower tumor growth kinetics and lower mitotic index, allowing for longer treatment windows and better modeling of early-stage tumor biology.
Tumors grow in a well-circumscribed, non-invasive manner and do not metastasize under standard experimental conditions. The model supports serial tumor volume measurement and endpoint tissue collection for pharmacodynamic and histological analyses. Because of its stable growth and epithelial integrity, RT112/84 is particularly well suited to testing agents that require prolonged exposure or mechanistic assessment over time.
While primarily employed in subcutaneous xenograft studies, RT112/84 has also been adapted for orthotopic bladder models using intravesical instillation or direct bladder wall injection, though these techniques require highly controlled microsurgical environments. The lack of spontaneous metastasis makes this model less applicable for studies focused on dissemination, but ideal for investigating tumor initiation, differentiation maintenance, and FGFR3 inhibition.
Request a Custom Quote for RT112/84 Xenograft ModelHistopathology and Immunohistochemical Profile
Tumors generated from RT112/84 xenografts exhibit a moderately differentiated transitional cell carcinoma morphology. Histologically, the tumors display broad sheets and nests of polygonal cells with moderate nuclear atypia, abundant cytoplasm, and distinct intercellular borders. The presence of cohesive epithelial architecture and minimal necrosis further supports the model’s utility for studying non-invasive or early-stage disease.
Immunohistochemical analysis reveals strong membranous expression of E-cadherin and cytokeratin 20, consistent with urothelial epithelial lineage. FGFR3 staining is diffusely cytoplasmic and membranous, confirming the relevance of the model for FGFR3-targeted pharmacology. Ki-67 proliferation index typically ranges from 20–35%, indicating a moderate growth rate suitable for longer-duration studies.
Additional staining for uroplakin II confirms the differentiated state of the tumor epithelium, while nuclear GATA3 and FOXA1 staining supports luminal subtype classification. Low expression of mesenchymal markers such as vimentin or N-cadherin and absence of MMP-9 signal a lack of EMT transition, reaffirming the model’s stable epithelial character.
The model demonstrates mild stromal infiltration and low vascular density, as indicated by CD31 staining, which correlates with its indolent behavior in vivo and non-metastatic phenotype.
Preclinical Applications and Drug Response
The RT112/84 xenograft model provides a reliable platform for evaluating targeted therapies against FGFR3, including small-molecule FGFR inhibitors, receptor-blocking antibodies, and combination strategies with downstream pathway inhibitors (e.g., PI3K or MEK inhibitors). The high and stable expression of wild-type FGFR3 enables robust pharmacodynamic measurement of drug target engagement, pathway suppression, and feedback activation.
Due to its non-mutated TP53 and preserved apoptotic machinery, RT112/84 responds predictably to DNA-damaging agents such as mitomycin C and low-dose cisplatin, making it suitable for modeling chemotherapeutic responses in less aggressive bladder tumors. The model has been employed in studies of differentiation therapy, where agents such as retinoic acid or HDAC inhibitors are tested for their ability to maintain or restore epithelial phenotype and suppress progression.
Unlike highly invasive models, RT112/84 offers limited insight into metastasis but provides significant translational value for drug development targeting early tumorigenic events, FGFR3-driven signaling, or maintenance of urothelial differentiation. It is frequently used in head-to-head comparisons of drug response across molecular subtypes, contributing to personalized therapy optimization for luminal-type bladder cancer.
Request This Model
To request the RT112/84 xenograft model for your preclinical studies or inquire about customization options such as orthotopic implantation, bioluminescent tagging, or extended-duration efficacy studies, please use the quote request form below. Our team is available to assist with study design, pricing, and integration into your development pipeline.
Request a Custom Quote for RT112/84 Xenograft Model