
Leverage an Extensive Portfolio of Fully Characterized PDX Models
Patient-derived xenograft (PDX) systems represent one of the most clinically relevant platforms for evaluating therapeutic efficacy in oncology research. Because these models originate directly from patient tumors and are never cultured in vitro, they maintain the full biological complexity, heterogeneity, and diversity seen in human cancers.
PDX studies provide a reliable and highly predictive indication of how a therapeutic candidate is likely to perform long before advancement into costly clinical development.
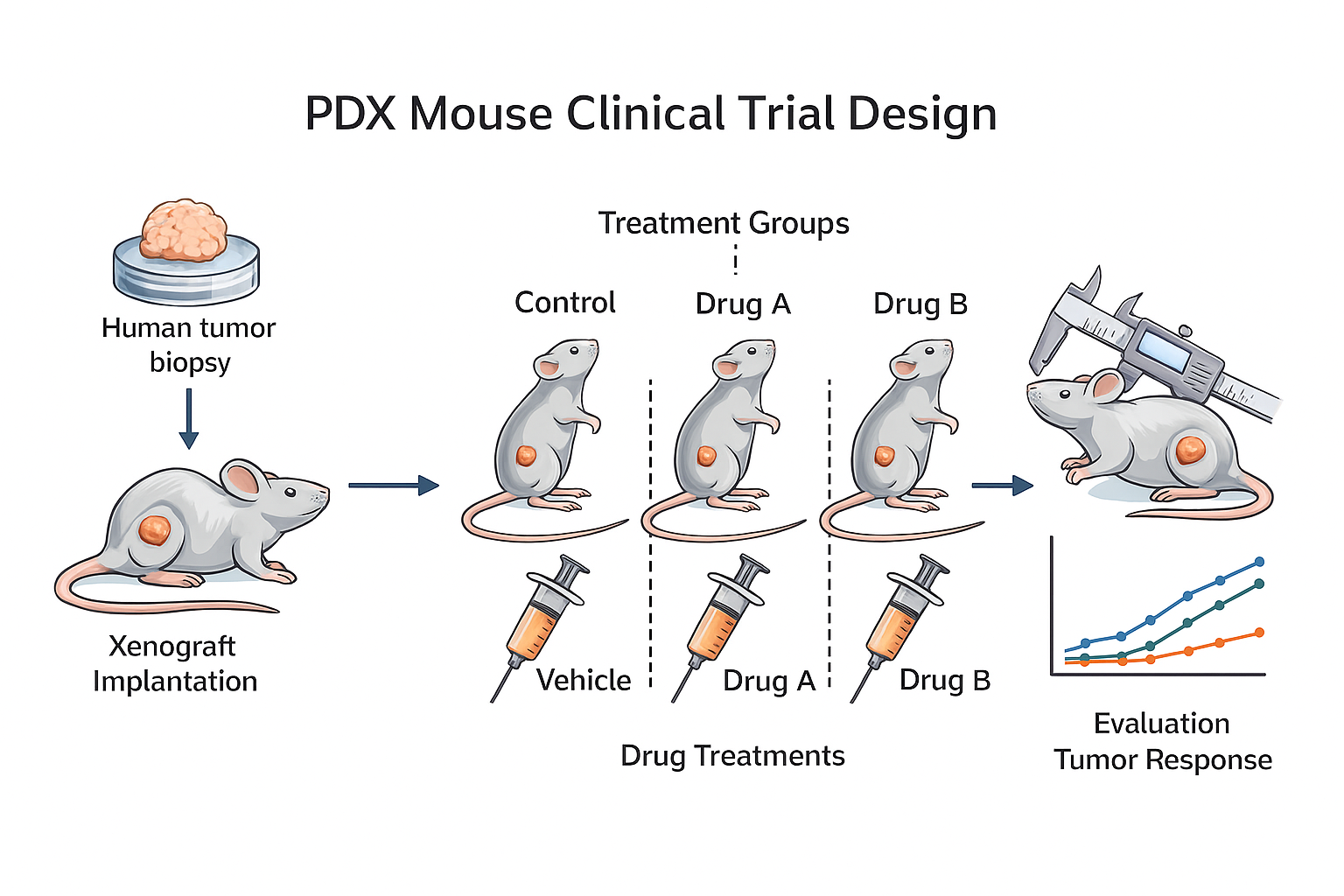
PDX Mouse Clinical Trial
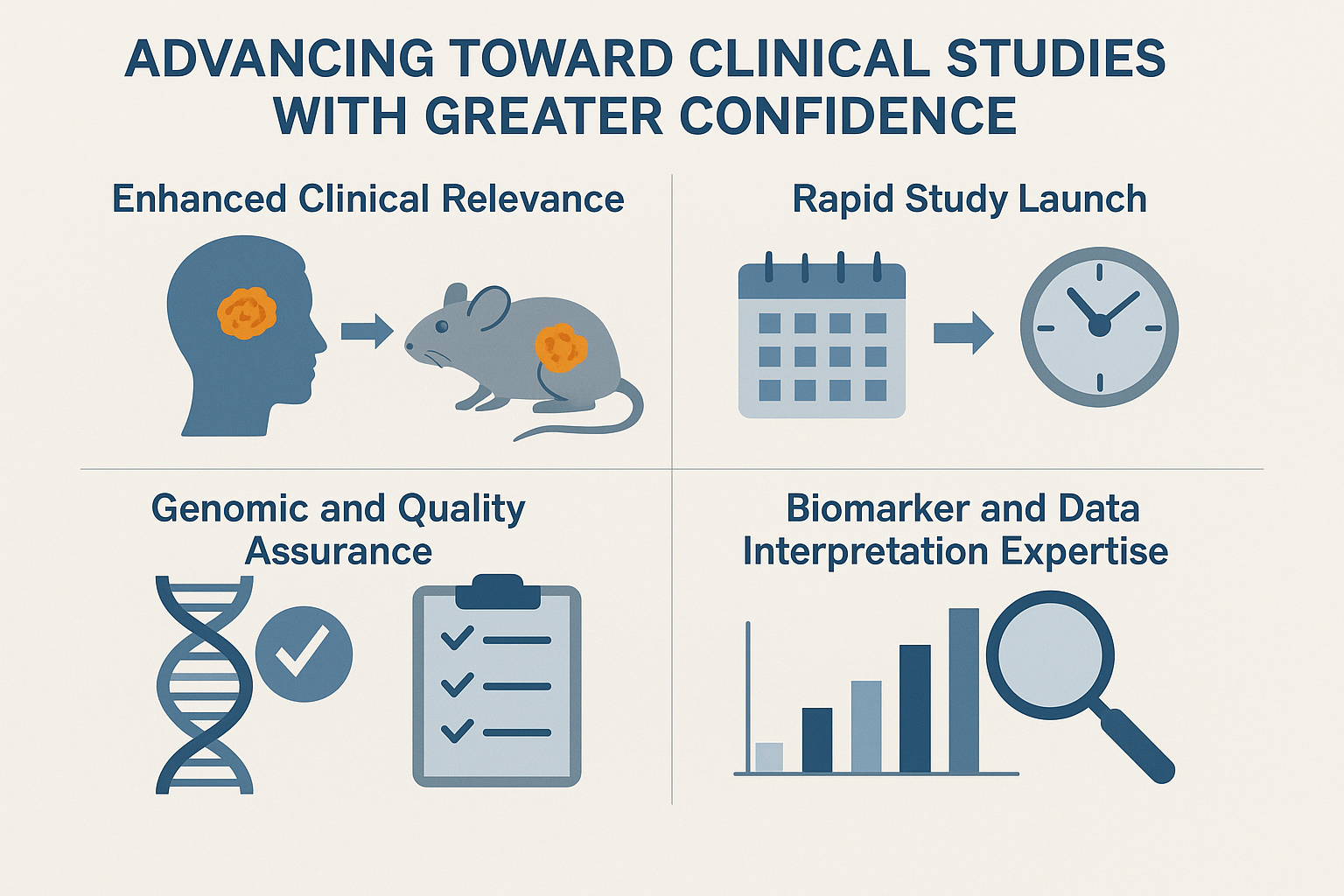
Advancing Toward Clinical Studies with Greater Confidence
PDX mouse clinical trials provide a powerful framework for oncology drug development, offering several key advantages:
- Enhanced Clinical Relevance: Because PDX models preserve the genetic, phenotypic, and architectural features of the original patient tumors, they more accurately represent human cancer biology. This leads to stronger prediction of therapeutic response before entering clinical trials.
- Rapid Study Launch: With a robust selection of live, ready-to-engraft models, we offer accelerated project initiation and shorter turnaround times for urgent development programs.
- Genomic and Quality Assurance: All models undergo stringent identity and quality assessments supported by next-generation sequencing–based QC workflows, ensuring reliability and preventing model drift or contamination.
- Biomarker and Data Interpretation Expertise: Our analytical framework supports biomarker evaluation and high-level interpretation of response data, even within complex or heterogeneous tumor environments.
- Integrated Bioinformatics Support: Our team provides guidance from study design through data analysis, ensuring that each PDX mouse clinical trial generates actionable translational insights.
Expertise and Capabilities at Xenograft.org
Xenograft.org provides comprehensive support for PDX mouse clinical trial–style studies, helping streamline translation from preclinical discovery to early clinical planning.
- Extensive Model Coverage: Our curated Xenograft Model Database includes approximately 300 well-characterized PDX models, representing diverse patient-derived tumor types and capturing real-world heterogeneity.
- Rapid Study Launch: With a robust selection of live, ready-to-engraft models, we offer accelerated project initiation and shorter turnaround times for urgent development programs.
- Genomic and Quality Assurance: All models undergo stringent identity and quality assessments supported by next-generation sequencing–based QC workflows, ensuring reliability and preventing model drift or contamination.
- Biomarker and Data Interpretation Expertise: Our analytical framework supports biomarker evaluation and high-level interpretation of response data, even within complex or heterogeneous tumor environments.
- Integrated Bioinformatics Support: Our team provides guidance from study design through data analysis, ensuring that each PDX mouse clinical trial generates actionable translational insights.
End-to-End Support for Translational Oncology
Xenograft.org offers an end-to-end solution that mirrors clinical trial conditions within a preclinical setting—improving predictive accuracy, reducing development risks, and accelerating decision-making. With extensive experience in PDX research, our platform serves as a reliable partner throughout the critical stages from preclinical testing to clinical advancement.