OE19 Xenograft Model Overview
The OE19 xenograft model is derived from a human esophageal adenocarcinoma cell line established from the primary tumor of a 72-year-old male patient. Representing a histological subtype strongly associated with Barrett’s esophagus and chronic gastroesophageal reflux, OE19 is widely used in preclinical studies of esophageal adenocarcinoma (EAC). The model demonstrates robust tumorigenicity, stable in vivo growth kinetics, and key molecular alterations commonly observed in human EAC, including ERBB2 (HER2) amplification. These features make OE19 xenografts a clinically relevant platform for evaluating HER2-targeted therapies, chemotherapeutic regimens, and combination treatments that address resistance mechanisms in gastroesophageal malignancies.
Request a Custom Quote for OE19 Xenograft ModelBiological and Molecular Characteristics
OE19 cells display epithelial morphology and retain molecular characteristics of well-differentiated esophageal adenocarcinoma. The cell line is HER2-positive due to ERBB2 gene amplification, a defining trait of a subset of EACs that predicts response to HER2-targeted agents such as trastuzumab and lapatinib. OE19 cells are TP53 mutant and express high levels of cytokeratin 8/18 (CK8/18), epithelial cell adhesion molecule (EpCAM), and E-cadherin, confirming their glandular epithelial origin. The model is microsatellite stable (MSS) and shows no activating KRAS or BRAF mutations, supporting its utility in HER2-driven disease contexts without confounding RAS/RAF-mediated resistance. Its expression of MUC1 and CDH1 further reinforces the EAC phenotype and provides opportunities for biomarker development and lineage-specific therapy evaluation.
| Characteristic | OE19 Cell Line Profile |
|---|---|
| Tissue of Origin | Esophageal adenocarcinoma (primary) |
| HER2 (ERBB2) Status | Amplified and overexpressed |
| KRAS/BRAF Status | Wild-type |
| TP53 Status | Mutated |
| MSI Status | Microsatellite stable (MSS) |
| Differentiation Markers | CK8/18, EpCAM, E-cadherin, MUC1, CDH1 |
In Vivo Model Development and Tumorigenicity
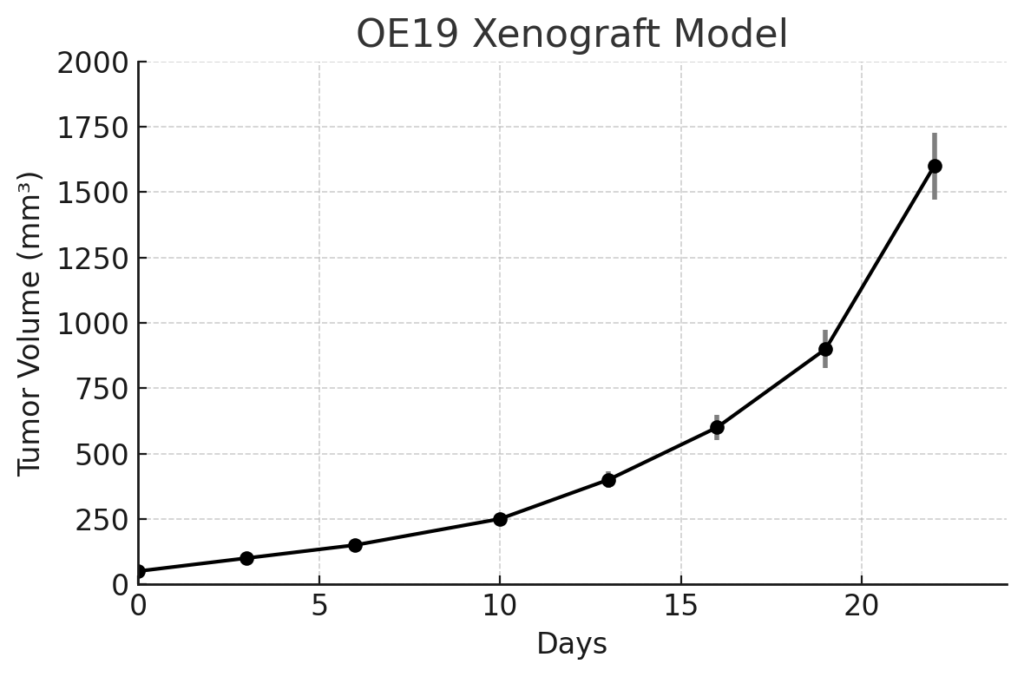
OE19 xenografts are generated via subcutaneous injection of cultured cells into immunodeficient mice, including athymic nude or NOD/SCID strains. Tumor take rates are high, with palpable masses forming within 7–10 days and volumes of 700–900 mm³ achieved by 4 to 5 weeks post-implantation. OE19 tumors grow in a uniform, nodular pattern with moderate vascularization, facilitating reproducible therapeutic studies. The model supports both monotherapy and combination treatment regimens, especially those involving HER2 inhibition, platinum-based chemotherapy, or immune checkpoint blockade. Its genetically defined HER2-driven tumorigenesis enables clear assessment of targeted drug response and resistance dynamics in esophageal adenocarcinoma.
Request a Custom Quote for OE19 Xenograft ModelHistopathology and Immunohistochemical Profile
Histological analysis of OE19 xenografts reveals moderately differentiated glandular structures consistent with esophageal adenocarcinoma. Hematoxylin and eosin (H&E) staining demonstrates columnar epithelial cells with gland formation, prominent nucleoli, and eosinophilic cytoplasm. Immunohistochemistry confirms strong membranous HER2 overexpression, supporting the model’s role in HER2-targeted research. Additional staining for CK8/18 and EpCAM confirms epithelial lineage, while E-cadherin localization at cell–cell junctions reflects intact adherens junctions. p53 staining is nuclear and diffuse, consistent with mutant TP53 accumulation. The model’s morphological fidelity and biomarker profile enhance its translational relevance for evaluating therapeutic interventions in HER2-positive esophageal adenocarcinoma.
Preclinical Applications and Drug Response
The OE19 xenograft model is instrumental in evaluating HER2-directed therapies, including trastuzumab, lapatinib, neratinib, and newer antibody–drug conjugates. Its high HER2 expression and wild-type KRAS status make it particularly suitable for mechanistic studies of HER2 signaling and therapeutic resistance in EAC. The model also supports evaluation of chemotherapy combinations such as cisplatin/5-FU and taxane-based regimens, especially in the context of HER2 amplification. Moreover, OE19 is utilized in studies assessing immune checkpoint inhibitors and agents targeting tumor-associated antigens expressed in EAC. Its defined molecular profile, rapid in vivo growth, and translational accuracy make OE19 a cornerstone for therapeutic development in esophageal adenocarcinoma.
Request This Model
To incorporate the OE19 xenograft model into your esophageal cancer research or preclinical program, contact our scientific team to request model specifications, study support, and access to this clinically relevant HER2-positive adenocarcinoma system.
Request a Custom Quote for OE19 Xenograft Model