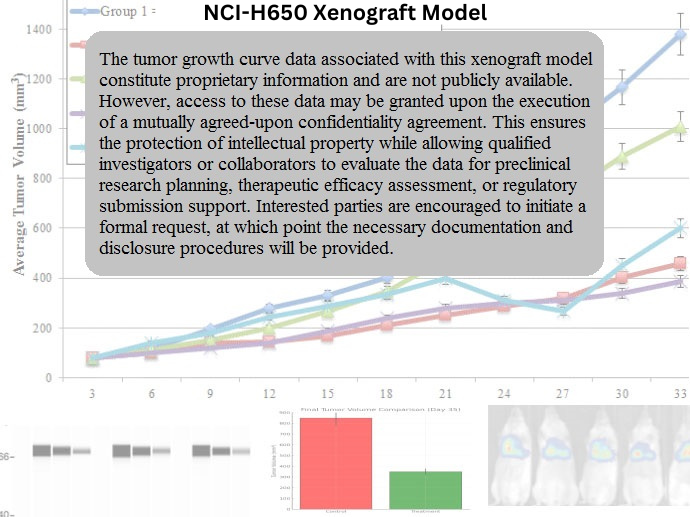
NCI-H650 Xenograft Model Overview
The NCI-H650 xenograft model is derived from a human large cell neuroendocrine carcinoma (LCNEC) of the lung, offering a valuable in vivo system for studying high-grade neuroendocrine tumors that share features with both small cell lung carcinoma (SCLC) and non-small cell lung cancer (NSCLC). Originating from a male patient, the NCI-H650 cell line exhibits a poorly differentiated neuroendocrine phenotype and forms tumors with moderate to rapid growth kinetics when implanted subcutaneously into immunodeficient mice. This model is especially relevant for evaluating novel therapies targeting neuroendocrine signaling, transcriptional regulation, and cell cycle dysregulation in aggressive pulmonary carcinomas that are resistant to conventional treatment strategies.
Request a Custom Quote for NCI‑H650 Xenograft ModelBiological and Molecular Characteristics
The NCI-H650 cell line displays hallmark features of neuroendocrine lung cancer, including high expression of synaptophysin, chromogranin A, and neuron-specific enolase (NSE). Genetically, it harbors loss-of-function mutations in both TP53 and RB1, a common molecular signature in high-grade neuroendocrine lung tumors. It lacks activating mutations in EGFR, KRAS, or ALK, allowing for targeted interrogation of non-epithelial oncogenic pathways. The model also exhibits amplification or overexpression of MYC family oncogenes, contributing to increased proliferation and transcriptional dependency. PD-L1 expression is typically low, and MHC class I presentation is diminished, creating a poorly immunogenic tumor microenvironment. These features position the NCI-H650 xenograft as a relevant system for studying neuroendocrine lineage plasticity, therapeutic resistance, and immune evasion.
| Characteristic | Description |
|---|---|
| Tissue Origin | Human lung (large cell neuroendocrine carcinoma) |
| Key Alterations | TP53 and RB1 inactivation; MYC overexpression |
| Mutation Status | EGFR/KRAS/ALK wild-type |
| Neuroendocrine Markers | Synaptophysin+, Chromogranin A+, NSE+ |
| Immunophenotype | PD-L1 (low), MHC I (low/variable) |
In Vivo Model Development and Tumorigenicity
The NCI-H650 xenograft model is established via subcutaneous implantation into immunodeficient mice, including NOD/SCID and athymic nude strains. Tumors typically become palpable within 10–14 days and grow steadily to experimental volumes of 300–500 mm³ within five to six weeks. The model demonstrates reliable tumor take and growth consistency, making it suitable for evaluating therapeutic responses, resistance mechanisms, and pharmacokinetic/pharmacodynamic (PK/PD) correlations. It is particularly advantageous for testing agents that target cell cycle regulators (e.g., CDK or aurora kinase inhibitors), transcriptional dependencies (e.g., BET or CDK7 inhibitors), or DNA repair pathways, including PARP and ATR inhibitors. Additionally, the model supports combination studies involving chemotherapy and epigenetic reprogramming.
Request a Custom Quote for NCI‑H650 Xenograft ModelHistopathology and Immunohistochemical Profile
Histologic examination of NCI-H650 xenograft tumors reveals a poorly differentiated neuroendocrine carcinoma with solid nests of small to intermediate-sized cells, scant cytoplasm, and granular nuclear chromatin. Hematoxylin and eosin staining highlights a high mitotic index, apoptotic debris, and focal areas of necrosis. Immunohistochemical analysis confirms strong positivity for synaptophysin and NSE, with variable chromogranin A staining. ASCL1 expression is commonly observed, indicating neuroendocrine lineage commitment. Ki-67 proliferation index typically exceeds 70%, reflecting the tumor’s aggressive biology. Epithelial markers such as cytokeratin are variably expressed, and PD-L1 staining is low or absent, supporting the model’s use in immune-sensitization or immunotherapy resistance research.
Preclinical Applications and Drug Response
The NCI-H650 xenograft model is extensively used in preclinical studies aimed at developing therapies for high-grade neuroendocrine lung cancers, particularly those resistant to platinum-based chemotherapy. It is sensitive to agents that target DNA replication stress and mitotic checkpoints, including topoisomerase inhibitors, CHK1 inhibitors, and aurora kinase inhibitors. The model has also been employed in testing small-molecule inhibitors of transcriptional machinery such as CDK9 or BET bromodomain inhibitors, as well as in evaluating metabolic vulnerabilities introduced by MYC overexpression. Its low PD-L1 expression and defective antigen presentation capacity make it a strong candidate for immunotherapy enhancement strategies, including epigenetic priming or STING agonist combinations. This model is an essential tool for dissecting the molecular complexity and treatment resistance of large cell neuroendocrine carcinomas.
Request This Model
To request the NCI-H650 xenograft model for your preclinical studies, please use the form below. A customized quote and additional model specifications will be provided upon inquiry.
Request a Custom Quote for NCI‑H650 Xenograft Model