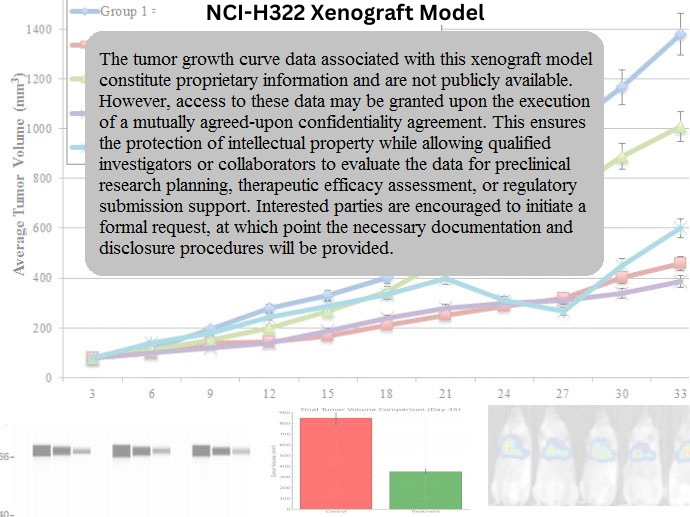
NCI-H322 Xenograft Model Overview
The NCI-H322 xenograft model is derived from a human non-small cell lung carcinoma (NSCLC) classified as a moderately differentiated adenocarcinoma. Originating from a 64-year-old male patient, this model retains hallmark features of lung adenocarcinomas driven by KRAS mutations and is frequently used in studies targeting RAS signaling and chemotherapy resistance. The NCI-H322 cell line exhibits well-differentiated epithelial characteristics, making it highly suitable for evaluating targeted therapeutics, epigenetic modulators, and immunomodulatory agents in lung cancer research. Its reproducible in vivo growth, combined with a distinct molecular profile, provides an essential tool for drug discovery in KRAS-driven lung adenocarcinoma.
Request a Custom Quote for NCI‑H322 Xenograft ModelBiological and Molecular Characteristics
NCI-H322 cells possess epithelial morphology and grow in adherent monolayers in vitro. The cell line harbors a KRAS G12C mutation, which constitutively activates the MAPK/ERK and PI3K/AKT signaling pathways. EGFR and TP53 genes are wild-type, allowing for the investigation of KRAS-specific oncogenic effects without interference from EGFR-driven pathways. NCI-H322 also expresses epithelial markers such as cytokeratins and E-cadherin, along with moderate levels of mucin-related genes like MUC1, reflecting partial glandular differentiation. These molecular features support the model’s use in studies focusing on KRAS G12C-selective inhibitors and in evaluating responses to MEK, PI3K, and ERK-targeted agents.
| Characteristic | NCI-H322 Cell Line Profile |
|---|---|
| Cancer Type | Non-small cell lung carcinoma (adenocarcinoma) |
| KRAS Status | Mutant (G12C) |
| EGFR/TP53 Status | Wild-type |
| Marker Expression | Cytokeratin⁺, E-cadherin⁺, MUC1⁺ |
| Growth Characteristics | Moderate proliferation, adherent epithelial monolayer |
In Vivo Model Development and Tumorigenicity
The NCI-H322 xenograft model is developed by subcutaneous injection of cultured cells into immunodeficient mice, typically athymic nude or NOD/SCID strains. Tumors begin to engraft within 7–10 days post-injection and reach volumes of 700–900 mm³ in approximately 5 weeks. The model demonstrates high reproducibility, consistent tumor take rates, and robust tumor progression, making it well-suited for longitudinal efficacy studies. NCI-H322 xenografts also exhibit moderate vascularization and stromal infiltration, enabling the evaluation of tumor–microenvironment interactions and drug penetration dynamics.
Request a Custom Quote for NCI‑H322 Xenograft ModelHistopathology and Immunohistochemical Profile
Histological examination of NCI-H322 xenografts reveals moderately differentiated adenocarcinoma with glandular structures, eosinophilic cytoplasm, and round-to-oval nuclei. The tumors often display acinar and papillary architectural patterns, characteristic of lung adenocarcinoma histotypes. Immunohistochemical analysis demonstrates strong expression of pan-cytokeratin and E-cadherin, confirming epithelial lineage. MUC1 staining is evident in luminal surfaces, and Ki-67 labeling indicates moderate to high proliferative activity. Phosphorylated ERK and AKT staining is observed, supporting constitutive RAS pathway activation due to the KRAS G12C mutation.
Preclinical Applications and Drug Response
The NCI-H322 xenograft model is a pivotal system for testing KRAS G12C-specific inhibitors and for evaluating agents that disrupt RAS-MAPK or PI3K-AKT signaling cascades. It serves as a valuable comparator in studies with other KRAS-mutant NSCLC models, especially those harboring G12D or G13D mutations. The model is also applicable for evaluating combination therapies involving immunotherapeutic agents, anti-angiogenics, and metabolic pathway modulators. Additionally, NCI-H322 supports investigations into resistance mechanisms against chemotherapy and small-molecule inhibitors, enabling development of more durable and personalized treatment strategies.
Request This Model
To explore the NCI-H322 xenograft model for your KRAS-targeted lung cancer research or preclinical drug testing pipeline, contact our scientific team for study design guidance, tumor growth data, and validated in vivo protocols supporting adenocarcinoma-focused therapeutic development.
Request a Custom Quote for NCI‑H322 Xenograft Model