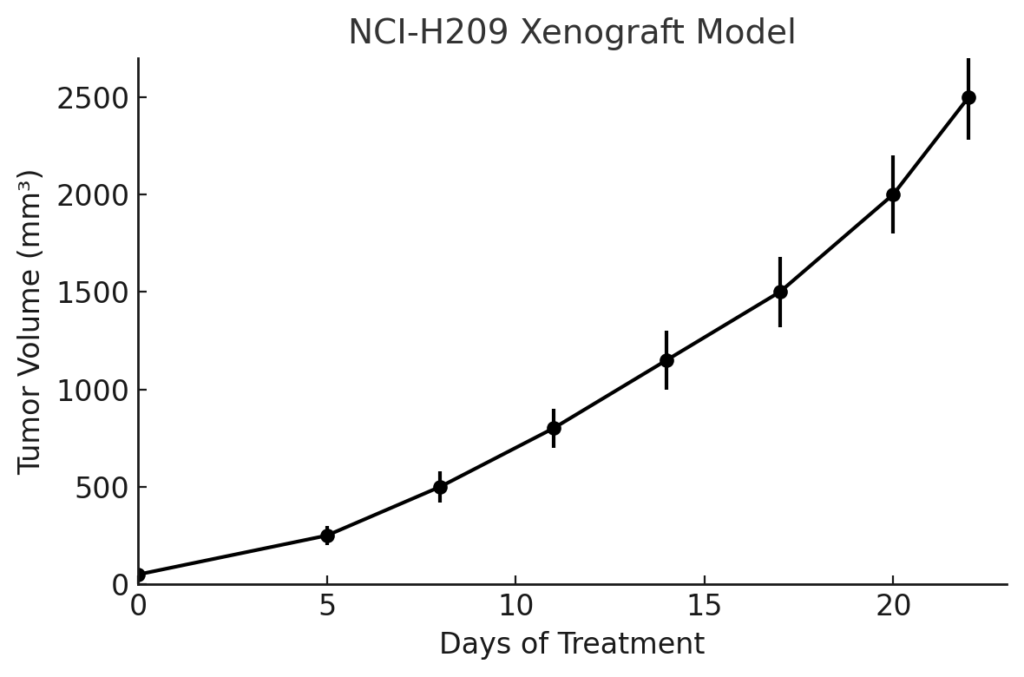
NCI-H209 Xenograft Model Overview
The NCI-H209 xenograft model is derived from a human small cell lung carcinoma (SCLC) and is widely utilized for preclinical research focused on neuroendocrine lung tumors. The parental NCI-H209 cell line was established from the lymph node metastasis of a male patient with extensive-stage SCLC. This model reflects the classical subtype of SCLC, exhibiting rapid tumor growth, high mitotic activity, and a robust neuroendocrine phenotype. It is particularly useful in evaluating the efficacy of DNA-damaging agents, apoptosis-sensitizing compounds, and novel therapeutic combinations targeting cell cycle regulation and chromatin remodeling. When implanted into immunodeficient mice, NCI-H209 forms highly cellular and aggressive tumors that recapitulate the histopathological features of clinical SCLC, making it an essential model for translational oncology studies.
Request a Custom Quote for NCI-H209 Xenograft ModelBiological and Molecular Characteristics
The NCI-H209 cell line is characterized by genetic alterations typical of classical SCLC, including biallelic inactivation of TP53 and RB1, which results in loss of cell cycle control and impaired DNA damage checkpoint responses. It expresses high levels of neuroendocrine markers such as synaptophysin, chromogranin A, and neuron-specific enolase (NSE), and demonstrates amplification of MYC, contributing to its aggressive phenotype and elevated proliferation. The model exhibits strong expression of BCL-2 and other anti-apoptotic regulators, making it suitable for studies evaluating BH3 mimetics and intrinsic apoptosis pathway disruption. The cell line is wild-type for KRAS and EGFR, and expresses low levels of PD-L1, providing a platform for immune sensitization studies despite its intrinsically low immunogenicity.
| Characteristic | Description |
|---|---|
| Tissue Origin | Human small cell lung carcinoma (lymph node metastasis) |
| Key Genetic Features | TP53 and RB1 inactivation, MYC amplification |
| Neuroendocrine Markers | Synaptophysin+, Chromogranin A+, NSE+ |
| Apoptotic Regulators | High BCL-2, survivin |
| Immunogenicity | Low PD-L1, intact MHC class I |
In Vivo Model Development and Tumorigenicity
The NCI-H209 xenograft model is established via subcutaneous injection of tumor cells into immunodeficient murine hosts, including athymic nude and NOD/SCID mice. Tumors are typically palpable within 7–10 days and progress rapidly, reaching target volumes of 400–700 mm³ within 3 to 4 weeks post-injection. This fast-growing tumor model is ideal for time-sensitive efficacy assessments and drug response profiling. The tumors exhibit high cellularity, well-defined morphology, and consistent vascularization, allowing for reproducible dosing and tissue sampling. Due to its rapid kinetics and neuroendocrine differentiation, the NCI-H209 model is particularly useful for simulating treatment cycles, investigating chemoresistance, and evaluating late-stage combination regimens that mimic clinical relapse scenarios.
Request a Custom Quote for NCI-H209 Xenograft ModelHistopathology and Immunohistochemical Profile
Tumors formed from the NCI-H209 xenograft model exhibit histological features consistent with poorly differentiated neuroendocrine carcinoma. Hematoxylin and eosin staining reveals dense sheets of small round cells with hyperchromatic nuclei, high nuclear-to-cytoplasmic ratios, and frequent mitotic figures. Ki-67 immunostaining indicates a proliferation index exceeding 80%, reinforcing the model’s rapid tumor growth and aggressive biology. Immunohistochemical analysis demonstrates robust expression of synaptophysin and chromogranin A, validating its neuroendocrine lineage. Strong cytoplasmic expression of BCL-2 is consistently observed, and nuclear accumulation of p53 reflects mutational inactivation. Although PD-L1 expression is minimal, the presence of MHC class I supports immunomodulatory investigation in combination with sensitizing agents.
Preclinical Applications and Drug Response
The NCI-H209 xenograft model is an established system for testing first-line and experimental therapies in SCLC. It responds predictably to platinum-based agents such as cisplatin and carboplatin, and to topoisomerase inhibitors including etoposide and irinotecan. The model has been employed extensively in studies involving BCL-2 antagonists, PARP inhibitors, and epigenetic modulators targeting MYC-driven transcriptional programs. Its MYC amplification status enhances its utility in evaluating synthetic lethal interactions, including ATR/CHK1 inhibition. Additionally, the model is used in combination therapy designs that integrate DNA damage induction with apoptosis potentiation. Although baseline PD-L1 expression is low, strategies involving immune checkpoint inhibitors have been explored in tandem with immunogenic chemotherapy to assess immune priming potential.
Request This Model
To request the NCI-H209 xenograft model for your preclinical studies, please use the form below. A customized quote and additional model specifications will be provided upon inquiry.
Request a Custom Quote for NCI-H209 Xenograft Model