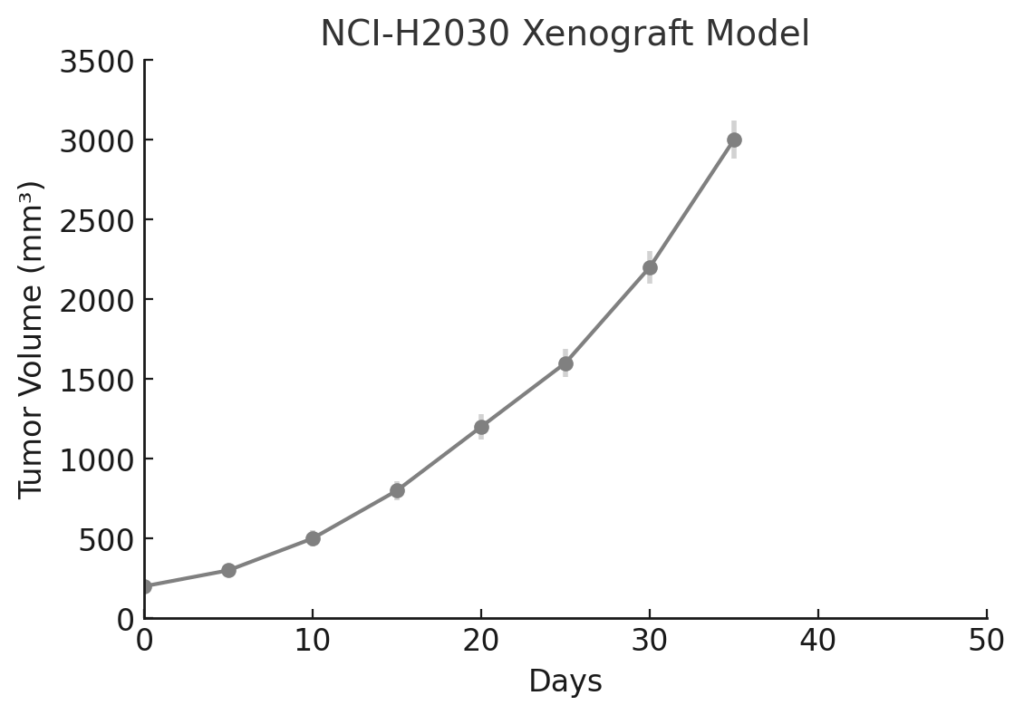
NCI-H2030 Xenograft Model Overview
The NCI-H2030 xenograft model is derived from a human lung adenocarcinoma and is extensively utilized in preclinical research for evaluating therapies targeting KRAS-mutant non-small cell lung cancer (NSCLC). Isolated from a male patient, the parental NCI-H2030 cell line harbors an activating KRAS mutation, placing it within a genetically defined subclass of lung cancer that exhibits resistance to EGFR-targeted therapies. When engrafted subcutaneously into immunodeficient mice, NCI-H2030 cells form solid tumors with consistent morphology and reproducible growth kinetics. This model is especially relevant for testing the efficacy of small-molecule inhibitors that target RAS signaling pathways, apoptosis regulators, and synthetic lethal partners of KRAS-driven oncogenesis. Its well-characterized molecular background and robust tumorigenicity make it a translationally relevant platform for therapy development in one of the most intractable subsets of NSCLC.
Request a Custom Quote for NCI-H2030 Xenograft ModelBiological and Molecular Characteristics
The NCI-H2030 cell line is defined by a KRAS^G12C mutation, a recurrent alteration found in a subset of NSCLC cases that confers constitutive activation of the MAPK and PI3K pathways. It is wild-type for EGFR, which makes it unresponsive to EGFR inhibitors and positions it as an ideal system for evaluating alternative therapeutic targets. The line exhibits an epithelial morphology and expresses cell adhesion markers such as E-cadherin and cytokeratin 7. Baseline expression of PD-L1 is low to moderate, and the presence of functional MHC class I molecules permits the exploration of immune-oncology strategies. NCI-H2030 cells also show upregulation of downstream signaling intermediates such as MEK1/2 and ERK1/2, providing a molecular framework conducive to combination therapies targeting both oncogenic signaling and apoptotic resistance.
| Characteristic | Description |
|---|---|
| Tissue Origin | Human lung (adenocarcinoma) |
| Key Genetic Features | KRAS^G12C mutant, EGFR wild-type, TP53 wild-type |
| Cell Morphology | Epithelial, adherent |
| Immunomarkers | CK7+, E-cadherin+, PD-L1 (low to moderate) |
| Oncogenic Pathways | MAPK, PI3K/AKT, RAS-dependent survival signaling |
In Vivo Model Development and Tumorigenicity
NCI-H2030 xenografts are established via subcutaneous injection into immunodeficient mice such as athymic nude or NOD/SCID strains. Tumor formation typically occurs within 10–14 days, with rapid progression to volumes of 400–600 mm³ within 4–5 weeks, depending on the cell dose and host strain. The model displays high take rates and reproducible tumor kinetics, allowing for controlled longitudinal efficacy evaluations and biomarker sampling. Its aggressive yet structurally organized tumor architecture supports drug distribution studies and pharmacokinetic profiling. The model’s compatibility with repeated dosing regimens, and its intact apoptotic machinery, make it suitable for evaluating both cytotoxic agents and targeted inhibitors. Engineered variants of NCI-H2030 are also available for studies incorporating imaging modalities or reporter gene-based therapeutic tracking.
Request a Custom Quote for NCI-H2030 Xenograft ModelHistopathology and Immunohistochemical Profile
Tumors generated from the NCI-H2030 xenograft model exhibit moderately differentiated histology with cohesive glandular structures and fibrous stroma. Hematoxylin and eosin staining reveals round to polygonal cells with moderate nuclear pleomorphism and frequent mitoses. The Ki-67 proliferation index ranges between 60% and 70%, indicative of a high-growth phenotype. Immunohistochemical staining confirms strong expression of cytokeratin 7 and membranous E-cadherin, supporting epithelial identity. PD-L1 expression is heterogeneous and focal, but sufficient to enable evaluation of checkpoint blockade strategies, particularly when combined with agents that increase tumor immunogenicity. The KRAS^G12C mutation is detectable by mutation-specific PCR or IHC-based surrogate markers, facilitating mechanistic studies and response monitoring.
Preclinical Applications and Drug Response
The NCI-H2030 xenograft model is frequently employed in preclinical studies targeting KRAS-mutant NSCLC, particularly for agents directed at the G12C allele. It demonstrates resistance to EGFR inhibitors, which highlights its role in non-oncogene-addicted therapeutic research. MEK inhibitors, ERK inhibitors, and emerging direct KRAS^G12C inhibitors such as sotorasib and adagrasib have shown variable efficacy in this model, especially when used in combination with BCL-2/BCL-XL inhibitors or immune-modulating agents. Its well-vascularized tumors support evaluation of drug delivery systems including liposomes, polymeric nanoparticles, and inhalable formulations. Given its intact p53 and low PD-L1 status, the model is well-suited for combination therapy studies focused on increasing immunogenicity or sensitizing tumors to immune checkpoint blockade.
Request This Model
To request the NCI-H2030 xenograft model for your preclinical studies, please use the form below. A customized quote and additional model specifications will be provided upon inquiry.
Request a Custom Quote for NCI-H2030 Xenograft Model