MDA-MB-436 Xenograft Model Overview
The MDA-MB-436 xenograft model originates from a human triple-negative breast cancer (TNBC) and is notable for its BRCA1-mutant genotype and mesenchymal phenotype. Derived from a pleural effusion of a 43-year-old Caucasian female patient with metastatic breast carcinoma, this model offers a clinically relevant system for studying homologous recombination deficiency, epithelial-to-mesenchymal transition (EMT), and therapeutic response to DNA-damaging agents and synthetic lethality-based treatments. MDA-MB-436 xenografts are particularly well suited for evaluating PARP inhibitors and investigating resistance mechanisms that develop in BRCA1-deficient tumors.
Unlike many other TNBC models, MDA-MB-436 is characterized by low proliferation rates and a spindle-shaped morphology, resembling basal B-like breast cancer. The xenograft recapitulates the complex interplay between tumor plasticity and impaired DNA repair, providing a powerful platform for assessing preclinical drug efficacy, biomarker validation, and tumor progression dynamics in the context of BRCA1 inactivation.
Request a Custom Quote for MDA-MB-436 Xenograft ModelBiological and Molecular Characteristics
The MDA-MB-436 cell line exhibits a well-defined BRCA1 mutation (5396+1G>A splice donor variant) that leads to loss of BRCA1 function and homologous recombination repair deficiency. It is triple-negative (ER-, PR-, HER2-) and possesses a mesenchymal-like gene expression profile with prominent EMT features. Expression of vimentin, N-cadherin, and fibronectin is high, while E-cadherin is absent, supporting its use in studies of cellular invasion and migration.
The model is p53-mutant and displays active PI3K/AKT signaling. In addition, expression of apoptosis-inhibiting proteins such as survivin and BCL-XL suggests an adaptive survival mechanism that operates despite ongoing DNA damage. Accumulation of γH2AX and defective RAD51 recruitment are key indicators of persistent genomic instability, especially following genotoxic stress.
| Characteristic | MDA-MB-436 Profile |
|---|---|
| Tumor Type | Triple-negative breast cancer (TNBC) |
| Origin | Pleural effusion, 43-year-old female |
| BRCA1 Status | Mutant (5396+1G>A splice site; non-functional) |
| TP53 Status | Mutant |
| Receptor Status | ER-, PR-, HER2- |
| EMT Markers | Vimentin+, N-cadherin+, Fibronectin+, E-cadherin– |
| Proliferation Index | Low to moderate |
| PI3K/AKT Signaling | Activated |
| RAD51 Localization | Cytoplasmic, non-functional |
| DNA Damage Indicators | High γH2AX, impaired HR |
This molecular profile positions MDA-MB-436 as a strong model for exploring BRCA1-deficient breast cancer progression, resistance, and response to targeted therapies.
In Vivo Model Development and Tumorigenicity
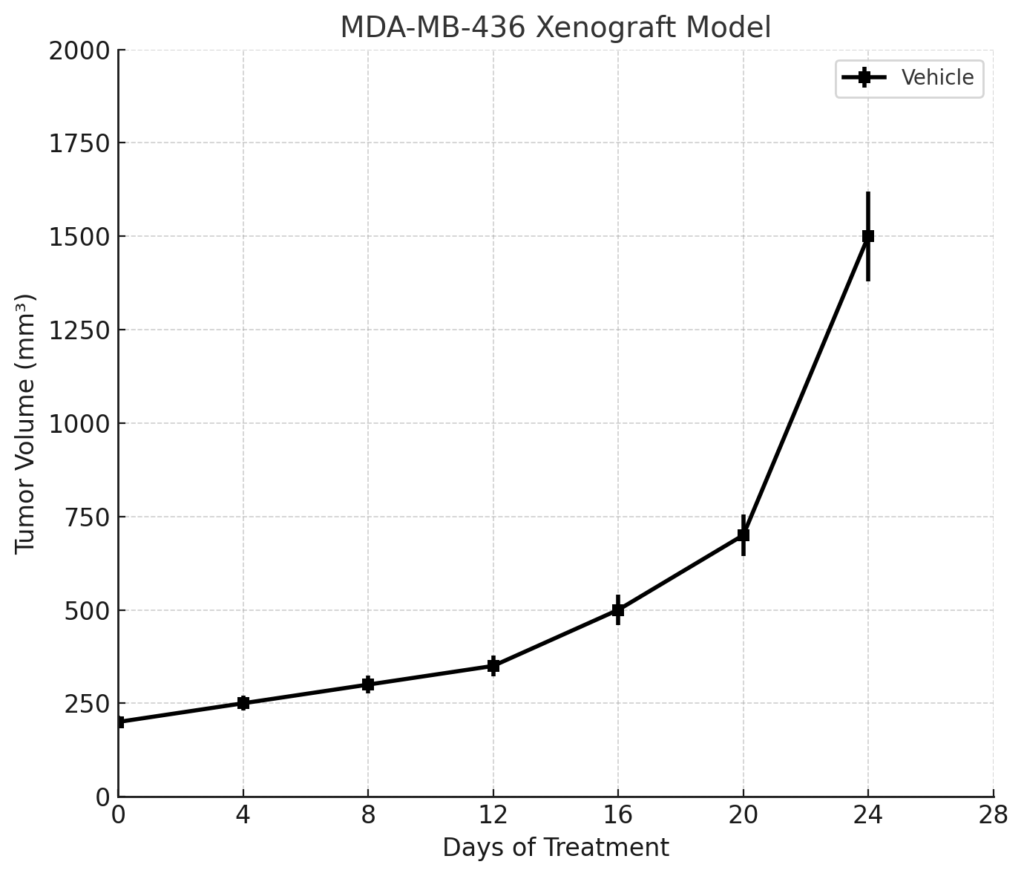
MDA-MB-436 xenografts are typically established in immunocompromised mice, most commonly NOD/SCID or athymic nude strains. Subcutaneous implantation of 5 × 10^6 to 1 × 10^7 cells in a Matrigel matrix results in tumors with moderate take rates (70–85%) and slower growth kinetics compared to other TNBC lines. Palpable tumors generally appear within 14–18 days and reach volumes of 800–1,200 mm³ within 4–6 weeks post-injection.
This model is also amenable to orthotopic implantation into the mammary fat pad, which allows for a more physiologically accurate tumor microenvironment and supports studies involving local invasion, extracellular matrix remodeling, and stromal interactions. Although spontaneous metastasis is infrequent, metastatic potential can be increased using genetic modifications or co-implantation with stromal components.
The relatively slow and steady growth rate makes MDA-MB-436 suitable for long-term treatment protocols, dose-escalation studies, and combination therapies, especially in the context of BRCA1 loss and low proliferation index.
Request a Custom Quote for MDA-MB-436 Xenograft ModelHistopathology and Immunohistochemical Profile
Histologically, MDA-MB-436 xenografts are composed of spindle-shaped tumor cells with irregular nuclear contours, prominent nucleoli, and a low-to-moderate mitotic index. The tumors often display loosely organized sheets or fascicles with interspersed fibrous stroma and limited necrosis. This architecture reflects the EMT-associated morphology seen in vitro.
Immunohistochemistry reveals strong expression of vimentin and N-cadherin, with loss of epithelial markers such as cytokeratin 8/18 and E-cadherin. Ki-67 staining ranges from 20–35%, consistent with the model’s modest proliferation rate. BRCA1 nuclear staining is absent, while γH2AX expression is elevated in both basal and treatment-exposed tumors, indicative of ongoing DNA damage.
Additional markers such as phospho-AKT, survivin, and BCL-XL are detected cytoplasmically, supporting the use of the model in preclinical testing of DNA damage response modulators and anti-apoptotic agents.
Preclinical Applications and Drug Response
The MDA-MB-436 xenograft model is particularly valuable for evaluating compounds that exploit BRCA1 deficiency. It has demonstrated sensitivity to PARP inhibitors such as olaparib and talazoparib, making it an essential system for synthetic lethality-based drug discovery. Moreover, it is highly suitable for testing platinum-based chemotherapeutics, including cisplatin and carboplatin.
Due to its EMT phenotype and active PI3K/AKT pathway, the model is also used to assess inhibitors targeting PI3K, AKT, mTOR, and downstream survival mechanisms. The partial mesenchymal character enables investigations into tumor cell plasticity, invasion, and resistance to standard chemotherapy.
MDA-MB-436 has been incorporated into studies of adaptive resistance, such as reversion of BRCA1 mutations, upregulation of error-prone DNA repair pathways, and epigenetic modulation. It is widely used in combination therapy protocols that pair DNA-damaging agents with checkpoint inhibitors (ATM, ATR, CHK1) or BCL-2 family antagonists.
Request This Model
To incorporate the MDA-MB-436 xenograft model into BRCA1-deficient breast cancer research, resistance modeling, or synthetic lethality screening, use the custom quote request link below. Support is available for both subcutaneous and orthotopic implantation, as well as drug dosing, imaging, and longitudinal tumor monitoring.
Request a Custom Quote for MDA-MB-436 Xenograft Model