Maver-1 Xenograft Model Overview
The Maver-1 xenograft model originates from a mantle cell lymphoma (MCL) cell line established from the peripheral blood of a patient with leukemic-phase MCL. This model represents the non-classical MCL subtype and provides an important platform for studying B-cell receptor (BCR) signaling, cyclin D1 overexpression, and resistance mechanisms in relapsed or refractory lymphoma. Unlike other MCL lines, Maver-1 harbors genetic features such as NOTCH1 mutations and elevated NF-κB pathway activity, enabling high translational relevance for targeted therapeutic research. When implanted subcutaneously into immunodeficient mice, Maver-1 cells produce tumors with moderate consistency and growth kinetics, suitable for pharmacologic studies involving both small molecules and antibody-based therapies.
Request a Custom Quote for Maver‑1 Xenograft ModelBiological and Molecular Characteristics
Maver-1 cells display a characteristic t(11;14)(q13;q32) chromosomal translocation, leading to overexpression of CCND1 (cyclin D1), a hallmark of mantle cell lymphoma. These cells are negative for CD10 and positive for pan-B-cell markers including CD19 and CD20, as well as CD5. Importantly, they exhibit constitutive activation of NF-κB signaling and often carry mutations in NOTCH1 and TRAF2, which modulate cell survival and immune evasion. The immunophenotype supports both surface immunoglobulin and MHC class II expression. Maver-1 also demonstrates low levels of PD-L1, but retains components of the antigen presentation machinery, allowing the model to be adapted for immunotherapy and checkpoint blockade evaluations.
| Characteristic | Description |
|---|---|
| Tissue Origin | Human mantle cell lymphoma (leukemic phase) |
| Key Alterations | t(11;14) CCND1 translocation, NOTCH1/TRAF2 mutations |
| Immunophenotype | CD19+, CD20+, CD5+, CD10–, MHC II+, PD-L1 (low) |
| Pathway Activation | Constitutive NF-κB and BCR signaling |
| Therapeutic Relevance | MCL therapy, BCR inhibition, NF-κB and CDK4/6 modulation |
In Vivo Model Development and Tumorigenicity
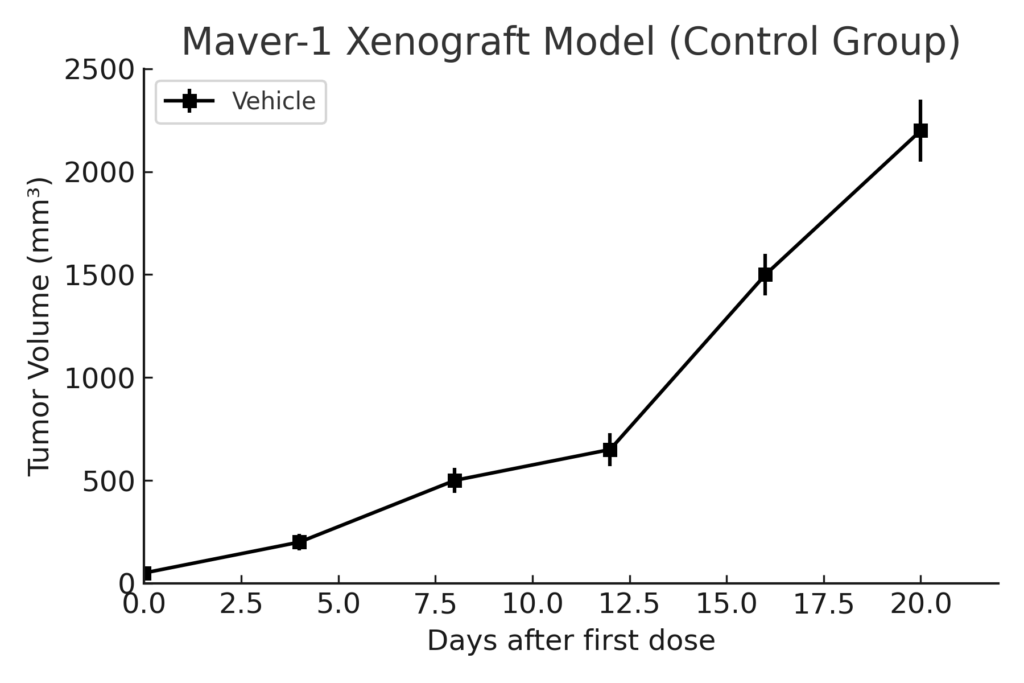
The Maver-1 xenograft model is developed by subcutaneous injection into immunodeficient mice such as NSG or NOD/SCID strains. Tumor formation is moderate in frequency, with palpable nodules typically observed by the third or fourth week post-implantation. Tumor volumes of 400–600 mm³ are generally reached in 6–8 weeks, allowing sufficient window for therapeutic intervention and endpoint evaluation. While tumor take rates are slightly lower than more aggressive lymphoid models, the biological fidelity of Maver-1 provides significant advantages in recapitulating relapsed or treatment-resistant MCL behavior. The model is especially useful in evaluating BCL2 inhibitors, BTK inhibitors, CDK4/6 antagonists, and immune modulators.
Request a Custom Quote for Maver‑1 Xenograft ModelHistopathology and Immunohistochemical Profile
Maver-1 xenografts exhibit histologic features characteristic of mantle cell lymphoma, including diffuse infiltration of small- to medium-sized lymphoid cells with irregular nuclear contours and scant cytoplasm. Hematoxylin and eosin staining demonstrates dense tumor cell populations with a low stromal component and variable mitotic activity. Immunohistochemical analysis reveals robust expression of cyclin D1, CD20, and BCL2. Nuclear p65 staining confirms active NF-κB signaling, and Ki-67 proliferation indices tend to be moderate, ranging from 30% to 50%. Despite low baseline PD-L1, preserved expression of HLA-DR and related MHC class II proteins supports its use in adaptive immune therapy research.
Preclinical Applications and Drug Response
The Maver-1 xenograft model is instrumental in the evaluation of targeted therapies for mantle cell lymphoma, particularly those aimed at disrupting BCR signaling and NF-κB pathway activation. It responds to BTK inhibitors such as ibrutinib and acalabrutinib, though with variable sensitivity reflective of clinical resistance profiles. CDK4/6 inhibitors like palbociclib show efficacy due to cyclin D1 overexpression, and BCL2 inhibitors (e.g., venetoclax) provide further avenues for apoptosis induction. The model is also suited for testing experimental antibody-drug conjugates, bispecific antibodies, and agents targeting NOTCH signaling, making it a valuable tool for translational MCL research.
Request This Model
To request the Maver-1 xenograft model for your preclinical studies, please use the form below. A customized quote and additional model specifications will be provided upon inquiry.
Request a Custom Quote for Maver‑1 Xenograft Model