LS411N Xenograft Model Overview
The LS411N xenograft model is established from a human colorectal adenocarcinoma cell line originally derived from the primary colon tumor of a 73-year-old male patient. This model is notable for its robust epithelial morphology, moderate tumorigenicity, and genetic profile consistent with microsatellite stability and chromosomal instability (CIN). LS411N cells are widely employed in preclinical studies that focus on epithelial-mesenchymal transition (EMT), Wnt/β-catenin signaling, and chemotherapeutic sensitivity in microsatellite-stable (MSS) colorectal cancer. The LS411N xenograft provides a reproducible platform for evaluating both monotherapies and combination regimens, particularly in the context of EGFR pathway responsiveness and oncogene-driven resistance. Its moderate growth kinetics and preserved histologic features make it a suitable model for longitudinal therapy studies and molecular biomarker validation.
Request a Custom Quote for LS411N Xenograft ModelBiological and Molecular Characteristics
LS411N cells are adherent and display well-defined epithelial morphology, with features such as tight junction formation and expression of E-cadherin. The line is characterized by a KRAS G13D mutation, which imparts resistance to EGFR inhibitors but retains susceptibility to downstream MAPK pathway inhibition. TP53 is also mutated, disrupting the canonical DNA damage response pathway. The cell line is microsatellite stable and exhibits chromosomal instability, consistent with the majority of sporadic colorectal cancers. It expresses moderate levels of carcinoembryonic antigen (CEA) and maintains active Wnt/β-catenin signaling, with partial nuclear localization of β-catenin. These molecular traits make LS411N a valuable system for interrogating resistance to receptor tyrosine kinase inhibitors, testing synthetic lethality strategies, and modeling epithelial colorectal cancer subtypes.
| Characteristic | LS411N Cell Line Profile |
|---|---|
| Tissue of Origin | Colorectal adenocarcinoma (primary) |
| KRAS Status | Mutant (G13D) |
| TP53 Status | Mutated |
| MSI Status | Microsatellite stable (MSS) |
| Wnt Signaling | Active, β-catenin partially nuclear |
| CEA Expression | Moderate |
In Vivo Model Development and Tumorigenicity
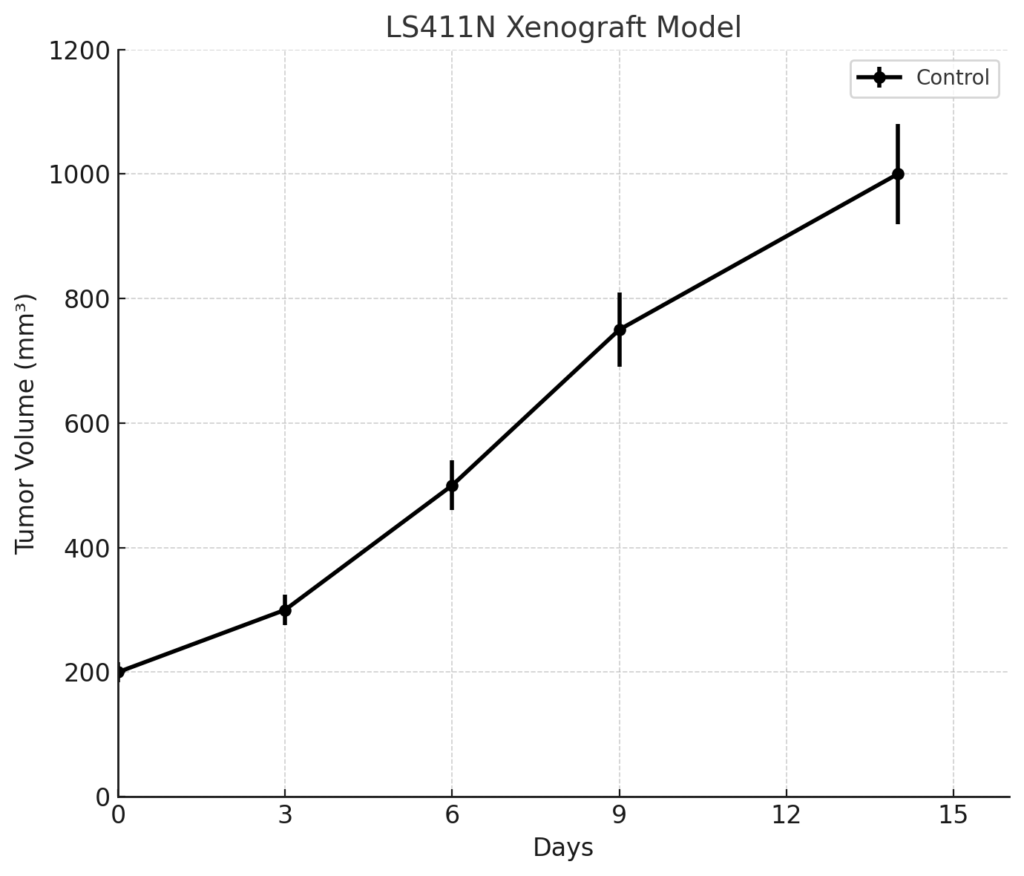
The LS411N xenograft is generated via subcutaneous injection of cultured cells into immunodeficient mice such as athymic nude or NOD/SCID strains. Tumor engraftment occurs within 10 to 14 days post-implantation, with steady volumetric progression over a 5- to 6-week period. Tumor volumes typically reach 700–900 mm³ in untreated controls, allowing for clear differentiation of treatment responses. The moderate proliferation rate and stable histology of LS411N tumors permit extended dosing schedules and pharmacodynamic sampling. The KRAS G13D mutation, in particular, provides a framework for testing rational drug combinations aimed at circumventing upstream receptor inhibition failures. The model’s tumorigenicity and molecular uniformity support its use in studies requiring precise control of genetic background and growth kinetics.
Request a Custom Quote for LS411N Xenograft ModelHistopathology and Immunohistochemical Profile
LS411N xenograft tumors exhibit moderately differentiated glandular structures with occasional luminal formation and central necrosis. Histological examination reveals epithelial features consistent with colorectal origin, including basally located nuclei and mucinous cytoplasm. Immunohistochemical staining confirms expression of CK20 and CEA, along with E-cadherin and EMA. β-catenin staining shows partial nuclear and membranous localization, consistent with active Wnt signaling. Mutant p53 protein accumulation is readily detectable by IHC, reflecting dysfunctional tumor suppressor activity. Tumors generally lack significant stromal infiltration or vascular heterogeneity, contributing to experimental consistency across cohorts. These histological and molecular characteristics validate LS411N as a clinically relevant model for studying MSS colorectal adenocarcinoma.
Preclinical Applications and Drug Response
The LS411N xenograft model is particularly effective for testing therapies in KRAS-mutant MSS colorectal cancer. Its resistance to EGFR-targeted agents like cetuximab is consistent with clinical outcomes in patients harboring KRAS G13D mutations, allowing for preclinical assessment of pathway inhibitors that act downstream of RAS. MEK and ERK inhibitors have shown antitumor activity in this model, particularly when combined with PI3K or mTOR inhibitors to address compensatory survival signaling. The model’s chromosomal instability and functional Wnt signaling pathway also support studies of therapeutic interventions targeting cell cycle regulators and transcriptional co-activators. Its consistent growth kinetics and preserved histologic phenotype make LS411N an optimal system for longitudinal efficacy studies, resistance profiling, and biomarker validation in colorectal cancer pipelines.
Request This Model
To incorporate the LS411N xenograft model into your preclinical program, contact our scientific team to discuss study objectives and request access to this genetically defined and translationally relevant colorectal cancer model.
Request a Custom Quote for LS411N Xenograft Model