LS1034 Xenograft Model Overview
The LS1034 xenograft model is derived from a human colorectal adenocarcinoma cell line that was initially established from a metastatic lesion in the lymph nodes of a 60-year-old female patient with advanced colon cancer. This model is widely utilized in preclinical studies focused on colorectal cancer, especially for testing therapeutic efficacy in metastatic disease. The LS1034 xenograft model is characterized by its ability to form primary tumors as well as secondary lesions, often in the liver and lungs, making it an ideal tool for evaluating the effectiveness of treatments against metastatic progression. The model’s stable growth, predictable tumor formation, and high metastatic potential make it a valuable resource for investigating new drugs, combination therapies, and immunotherapeutic strategies.
Request a Custom Quote for LS1034 Xenograft ModelBiological and Molecular Characteristics
LS1034 cells exhibit epithelial morphology and form well-polarized monolayers in vitro. The cell line is microsatellite stable (MSS) and carries a wild-type KRAS, BRAF, and NRAS profile, making it responsive to EGFR-targeted therapies such as cetuximab and panitumumab. TP53 is mutated, which compromises apoptotic signaling and allows for the evaluation of DNA-damaging agents, as well as therapies that exploit synthetic lethality. LS1034 cells express moderate levels of carcinoembryonic antigen (CEA) and cytokeratin 20 (CK20), confirming their colorectal origin. Wnt/β-catenin signaling is active, with β-catenin found both in the cytoplasm and at the membrane, indicating intact signaling. These molecular traits render LS1034 a robust model for studying the effects of EGFR inhibition, chemotherapeutic agents, and combination therapies in colorectal cancer with metastatic potential.
| Characteristic | LS1034 Cell Line Profile |
|---|---|
| Tissue of Origin | Colorectal adenocarcinoma (metastatic) |
| KRAS/BRAF/NRAS Status | Wild-type |
| TP53 Status | Mutated |
| MSI Status | Microsatellite stable (MSS) |
| Differentiation Markers | CK20, CEA, E-cadherin |
| Wnt Signaling | Active, β-catenin cytoplasmic/membranous |
In Vivo Model Development and Tumorigenicity
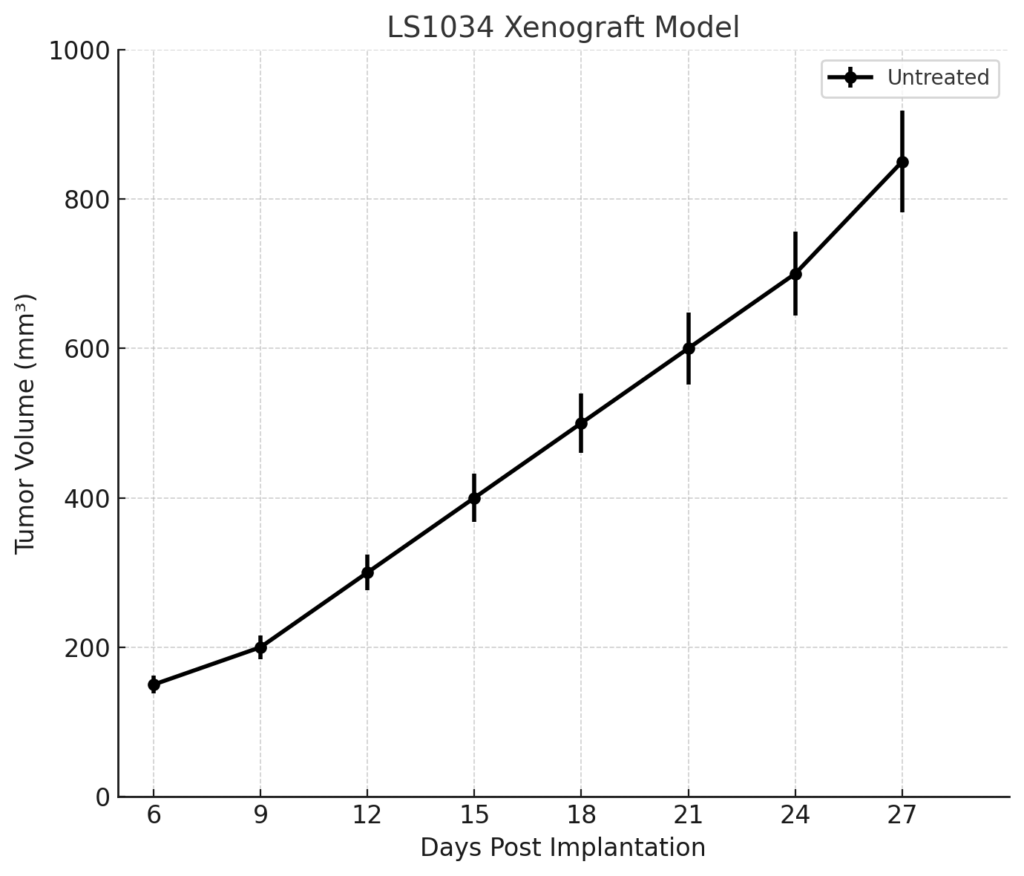
LS1034 xenografts are typically developed by subcutaneous or orthotopic injection of cultured cells into immunodeficient mouse strains, such as athymic nude or NOD/SCID mice. Tumor formation is reliable and occurs within 7–10 days post-inoculation, with tumors reaching volumes of 700–900 mm³ by day 28–35. The model exhibits high metastatic potential, with secondary tumors often forming in the liver and lungs. This characteristic makes LS1034 particularly valuable for studies focused on metastatic colorectal cancer and therapies targeting metastasis, invasion, and tumor microenvironment interactions. The consistent tumor growth kinetics and the ability to replicate metastasis provide a robust platform for long-term studies and multi-cycle therapeutic evaluations.
Request a Custom Quote for LS1034 Xenograft ModelHistopathology and Immunohistochemical Profile
Histologically, LS1034-derived xenografts exhibit moderately differentiated adenocarcinomas with glandular structures, mild mucin production, and regions of necrosis. Hematoxylin and eosin (H&E) staining highlights columnar epithelial cells with basal nuclei and clear cytoplasm, resembling the morphology of human colorectal carcinomas. Immunohistochemistry confirms positive staining for CK20 and CEA, indicating the colorectal origin of the tumor. E-cadherin expression is present at cell–cell junctions, supporting the epithelial nature of the tumor. β-catenin shows both cytoplasmic and membranous localization, reflecting active Wnt signaling. Mutant p53 is detected in the tumor nuclei, confirming the loss of normal apoptotic control. The ability of LS1034 xenografts to develop metastases, especially in the liver and lungs, makes them particularly useful for studying the mechanisms of metastasis and testing anti-metastatic therapies.
Preclinical Applications and Drug Response
The LS1034 xenograft model is highly relevant for evaluating therapies targeting metastatic colorectal cancer. Given its wild-type KRAS/BRAF status, the model is responsive to EGFR-targeted therapies such as cetuximab and panitumumab, making it a valuable platform for evaluating these agents in metastatic disease. The TP53 mutation in this model provides an opportunity to test DNA-damaging agents and therapies that exploit synthetic lethality. Additionally, LS1034 xenografts are ideal for studying metastasis and evaluating drugs designed to inhibit tumor migration, invasion, and angiogenesis. The model’s high metastatic potential and stable in vivo growth make it a robust system for testing combination therapies, immunotherapies, and novel drug candidates aimed at overcoming metastatic progression in colorectal cancer.
Request This Model
To incorporate the LS1034 xenograft model into your colorectal cancer research program, contact our scientific team for detailed model specifications, customized study designs, and access to this highly reproducible metastatic cancer model.
Request a Custom Quote for LS1034 Xenograft Model