KYSE-520 Xenograft Model Overview
The KYSE-520 xenograft model originates from a human esophageal squamous cell carcinoma (ESCC) cell line derived from a poorly differentiated primary tumor in a 67-year-old Japanese male patient. As one of the extensively characterized KYSE series lines, KYSE-520 offers strong translational value for preclinical studies of ESCC, particularly in investigating radiation sensitization, therapeutic resistance, and molecular drivers of squamous carcinogenesis. The model is noted for its aggressive tumor growth, histopathological consistency, and high tumor take rate in immunodeficient mice. With molecular alterations representative of high-grade ESCC, the KYSE-520 xenograft provides an effective platform for testing both standard therapies and novel experimental agents in squamous esophageal carcinoma.
Request a Custom Quote for KYSE-520 Xenograft ModelBiological and Molecular Characteristics
KYSE-520 cells possess classic squamous epithelial morphology and express high levels of cytokeratins typical of squamous lineage, including CK5/6 and CK14. The cell line is TP53 mutant, reflecting the frequent p53 dysregulation observed in ESCC, and also exhibits alterations in the PI3K/AKT pathway. It is wild-type for KRAS and BRAF, eliminating confounding RAS pathway resistance when studying targeted inhibitors. KYSE-520 demonstrates overexpression of EGFR and SOX2, the latter being a key transcription factor implicated in squamous cell stemness and oncogenesis. The model is microsatellite stable (MSS) and exhibits a strong epithelial signature, with additional expression of p63 and involucrin, supporting its utility for evaluating therapies directed at squamous-specific signaling, proliferation, and differentiation.
| Characteristic | KYSE-520 Cell Line Profile |
|---|---|
| Tissue of Origin | Esophageal squamous cell carcinoma (ESCC) |
| TP53 Status | Mutated |
| KRAS/BRAF Status | Wild-type |
| EGFR Expression | Overexpressed |
| Squamous Markers | CK5/6, CK14, p63, involucrin |
| SOX2 Status | Overexpressed |
In Vivo Model Development and Tumorigenicity
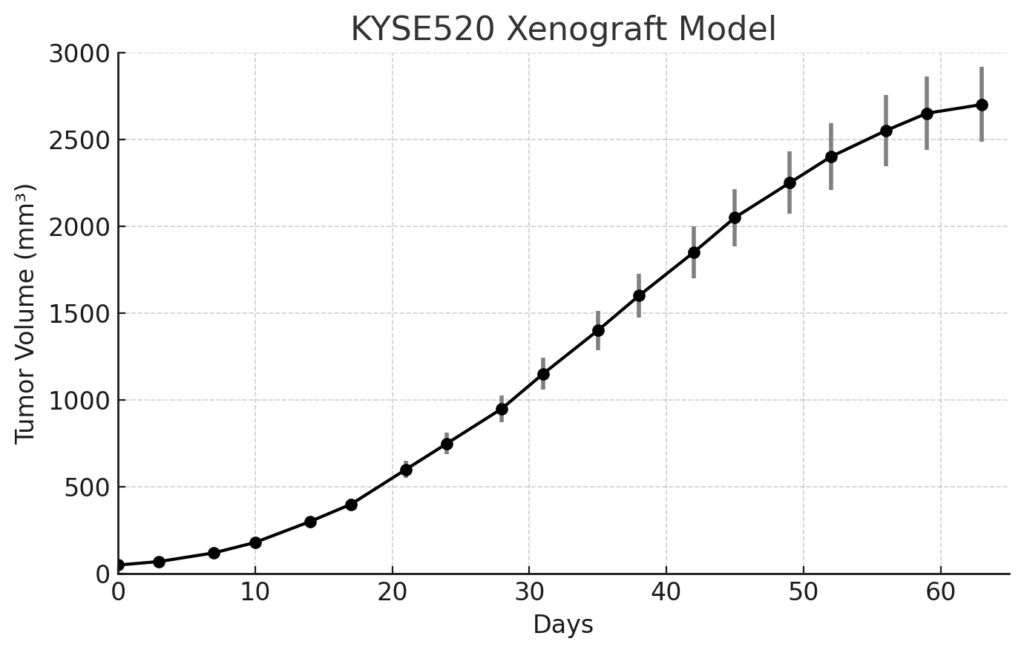
KYSE-520 xenografts are established by subcutaneous injection of cultured cells into immunodeficient mouse models such as athymic nude or NOD/SCID strains. Tumors typically engraft within 7 to 10 days post-implantation and grow rapidly, reaching experimental volumes of 700–900 mm³ in approximately 3 to 4 weeks. The model exhibits a high tumor incidence and uniform growth patterns, allowing for reliable replication of experimental conditions across cohorts. Its aggressive growth characteristics and high mitotic index make it suitable for evaluating antimitotic agents, radiation protocols, and combinations involving EGFR or PI3K pathway inhibitors. KYSE-520 xenografts also lend themselves to investigations of resistance to cytotoxic and molecularly targeted treatments in ESCC.
Request a Custom Quote for KYSE-520 Xenograft ModelHistopathology and Immunohistochemical Profile
Histologically, KYSE-520 xenografts exhibit features typical of poorly differentiated squamous cell carcinoma. Tumors consist of solid sheets and nests of pleomorphic epithelial cells with high nuclear-cytoplasmic ratios, hyperchromatic nuclei, and minimal keratinization. Hematoxylin and eosin (H&E) staining reveals focal necrosis and regions of active mitosis. Immunohistochemical analysis confirms strong and diffuse expression of squamous lineage markers, including CK5/6, p63, and SOX2. EGFR staining is membrane-localized, consistent with receptor overexpression. Nuclear p53 accumulation is frequently observed, reflecting the underlying TP53 mutation. These findings confirm the tumor’s squamous origin and aggressive phenotype, reinforcing its relevance for ESCC research.
Preclinical Applications and Drug Response
The KYSE-520 xenograft model is extensively used in ESCC-focused preclinical studies aimed at evaluating responses to EGFR inhibitors, radiation, chemotherapeutics, and targeted molecular agents. Its high EGFR expression and wild-type RAS status make it a rational model for studying EGFR-targeted therapies alone or in combination with DNA-damaging agents. The model is also ideal for testing inhibitors of the PI3K/AKT pathway, agents that target squamous transcriptional programs such as SOX2, and compounds modulating p53-dependent apoptosis. Additionally, KYSE-520 supports drug resistance modeling and biomarker development in squamous esophageal cancers, offering a robust system for translational research and therapy optimization.
Request This Model
To incorporate the KYSE-520 xenograft model into your esophageal cancer drug development or preclinical testing strategy, contact our scientific team to obtain full model specifications and initiate a custom-designed study tailored to your therapeutic goals.
Request a Custom Quote for KYSE-520 Xenograft Model