KP4 Xenograft Model Overview
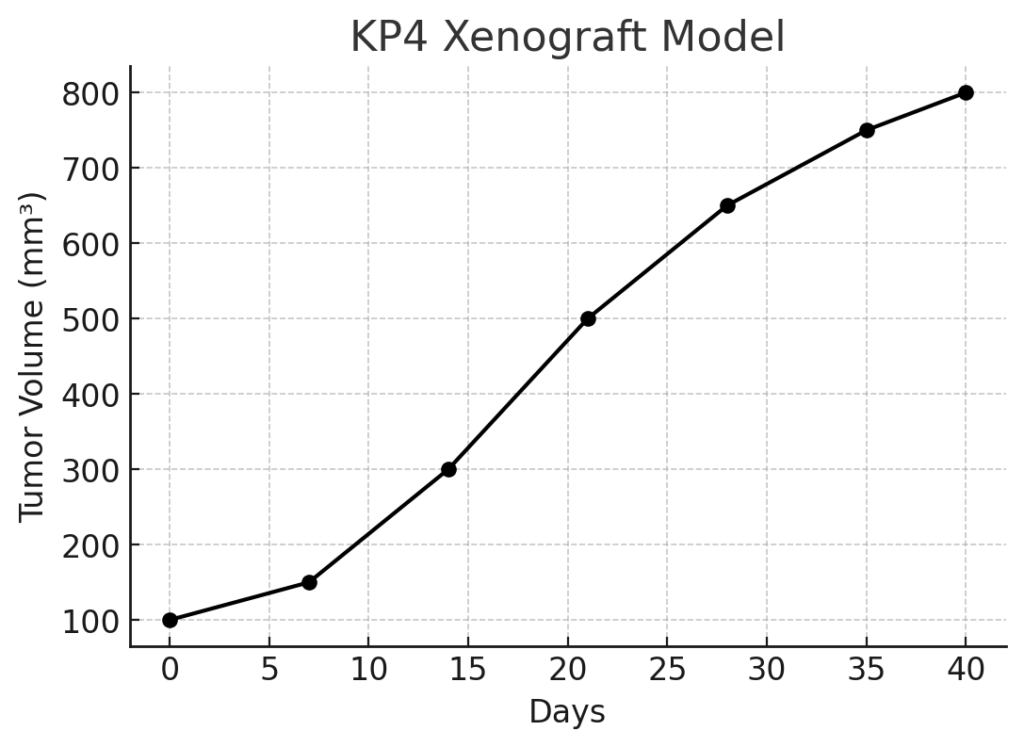
The KP4 xenograft model is derived from a human pancreatic cancer cell line, KP4, which was established from a patient with pancreatic ductal adenocarcinoma (PDAC). PDAC is one of the most aggressive and lethal cancers, characterized by late-stage diagnosis, rapid tumor growth, and a tendency to metastasize early to distant organs, including the liver, lungs, and peritoneum. The KP4 xenograft model is widely used in preclinical studies to investigate the biology of pancreatic cancer, its metastatic behavior, and to evaluate novel therapeutic strategies aimed at improving treatment outcomes. Given the aggressive nature of PDAC and its resistance to most treatments, the KP4 model is highly valuable for testing chemotherapy agents, targeted therapies, and immunotherapies.
Request a Custom Quote for KP4 Xenograft ModelBiological and Molecular Characteristics
KP4 cells are characterized by their epithelial origin and express several key markers typical of pancreatic cancer, including cytokeratins, epithelial membrane antigen (EMA), and the cancer-associated antigen CA19-9, which is often elevated in pancreatic cancer patients. The model exhibits a dysregulated KRAS signaling pathway, a hallmark of PDAC, which plays a critical role in tumor initiation, progression, and resistance to therapy. Additionally, KP4 cells show loss of the tumor suppressor gene p53, contributing to tumor cell survival and resistance to apoptosis. The model also exhibits high expression of matrix metalloproteinases (MMPs) that facilitate tumor invasion and metastasis. These molecular features make KP4 xenografts a relevant model for studying the tumor microenvironment, metastasis, and novel drug candidates targeting key signaling pathways in pancreatic cancer.
| Marker | Expression Level | Function |
|---|---|---|
| Cytokeratin | High | Epithelial cell marker |
| EMA | High | Epithelial membrane antigen |
| CA19-9 | Elevated | Pancreatic cancer biomarker |
| KRAS | Mutated | Oncogene involved in tumor initiation and progression |
| MMPs | High | Involved in tumor invasion and metastasis |
In Vivo Model Development and Tumorigenicity
The KP4 xenograft model is typically established by implanting KP4 cells into immunocompromised mice, such as NOD/SCID or NSG mice. Upon implantation, the cells rapidly proliferate, forming solid tumors that replicate the aggressive growth patterns of human pancreatic cancer. The tumors exhibit high cellularity, frequent necrosis, and significant vascularization, which are indicative of the rapid tumor growth and angiogenesis. The model is frequently used for evaluating the efficacy of chemotherapy agents such as gemcitabine, which is commonly used in the treatment of pancreatic cancer. However, due to the inherent resistance of pancreatic tumors to many treatment regimens, the KP4 model is also valuable for testing combination therapies, novel chemotherapy agents, and targeted therapies.
In addition to subcutaneous implantation, orthotopic implantation of KP4 cells into the pancreas of immunocompromised mice can be performed to replicate the natural site of tumor growth and metastasis. This orthotopic model allows for the study of pancreatic cancer’s local invasion and metastasis to distant organs, which is a key feature of PDAC. It also provides a more clinically relevant setting for evaluating the effects of treatments on both primary tumors and metastatic lesions.
Request a Custom Quote for KP4 Xenograft ModelHistopathology and Immunohistochemical Profile
Histopathological analysis of KP4 xenografts reveals tumors with a characteristic morphology of pancreatic ductal adenocarcinoma, including irregular glandular structures and areas of necrosis. The tumors are composed of pleomorphic cells with abundant eosinophilic cytoplasm and high mitotic activity. Immunohistochemical staining of KP4 xenografts shows high expression of cytokeratins and EMA, confirming the epithelial origin of the tumor. The tumors also show elevated levels of CA19-9, which is commonly used as a biomarker for pancreatic cancer diagnosis and monitoring. Additionally, KP4 xenografts exhibit a high degree of vascularization, assessed by CD31 staining, indicating the tumors’ reliance on angiogenesis for sustained growth. The tumors also express high levels of matrix metalloproteinases (MMPs), which are associated with the invasive potential of the cancer cells and their ability to metastasize.
Preclinical Applications and Drug Response
The KP4 xenograft model is extensively used to evaluate the efficacy of various therapeutic agents for pancreatic cancer. The model is particularly useful for testing chemotherapy agents, such as gemcitabine, which is the standard treatment for PDAC. However, due to the development of resistance to gemcitabine, the KP4 model is also valuable for testing novel drugs and combination therapies aimed at overcoming chemotherapy resistance. The model is frequently used to assess the effectiveness of targeted therapies that inhibit key signaling pathways involved in PDAC progression, such as KRAS, PI3K/AKT, and MAPK/ERK pathways. Additionally, the KP4 xenograft model is employed to evaluate immunotherapies, including immune checkpoint inhibitors and adoptive T-cell therapies, which have shown promise in clinical trials for various cancers, including pancreatic cancer.
In addition to drug testing, the KP4 model is used to study the tumor microenvironment, including interactions between tumor cells and stromal cells, immune cells, and the extracellular matrix. Understanding these interactions is critical for developing therapies that can target the tumor microenvironment and improve treatment efficacy.
Request This Model
To request the KP4 xenograft model for your preclinical studies, please use the form below. A customized quote and additional model specifications will be provided upon inquiry.
Request a Custom Quote for KP4 Xenograft Model