JHH-7 Xenograft Model Overview
The JHH-7 xenograft model is based on a human hepatocellular carcinoma (HCC) cell line derived from a Japanese male patient. Known for its high tumorigenicity and well-defined hepatocyte-like characteristics, JHH-7 serves as a robust in vivo model for studying liver cancer biology and evaluating targeted therapeutic agents. This cell line is p53 wild-type and exhibits a phenotype more closely aligned with differentiated hepatocytes compared to other HCC lines, making it particularly relevant for modeling hepatocellular carcinomas with preserved hepatic functionality. The JHH-7 xenograft model is well-suited for pharmacodynamic analyses, drug resistance profiling, and investigation of oncogenic signaling within a hepatic tumor microenvironment.
Request a Custom Quote for JHH‑7 Xenograft ModelBiological and Molecular Characteristics
JHH-7 cells express classic hepatocellular markers including albumin, alpha-fetoprotein (AFP), and hepatocyte nuclear factor 4 alpha (HNF4A), alongside cytokeratins CK8 and CK18. The cell line is notable for its functional expression of bile acid transporters and cytochrome P450 enzymes, supporting studies on hepatic drug metabolism and transporter-mediated pharmacokinetics. JHH-7 is wild-type for TP53, distinguishing it from many HCC lines that harbor common p53 mutations. Signaling pathways active in JHH-7 include Wnt/β-catenin, TGF-β, and PI3K/AKT, with moderate activity in ERK/MAPK signaling. Its differentiated features allow for mechanistic studies of hepatic carcinogenesis and treatment-induced hepatotoxicity.
| Characteristic | JHH-7 Cell Line Profile |
|---|---|
| Cancer Type | Hepatocellular carcinoma (HCC) |
| Species/Tissue of Origin | Human liver |
| Key Markers | AFP⁺, Albumin⁺, HNF4A⁺, CK18⁺ |
| TP53 Status | Wild-type |
| Active Signaling Pathways | Wnt/β-catenin, PI3K/AKT, TGF-β |
| Applications | Hepatic drug metabolism, pathway inhibition, HCC models |
In Vivo Model Development and Tumorigenicity
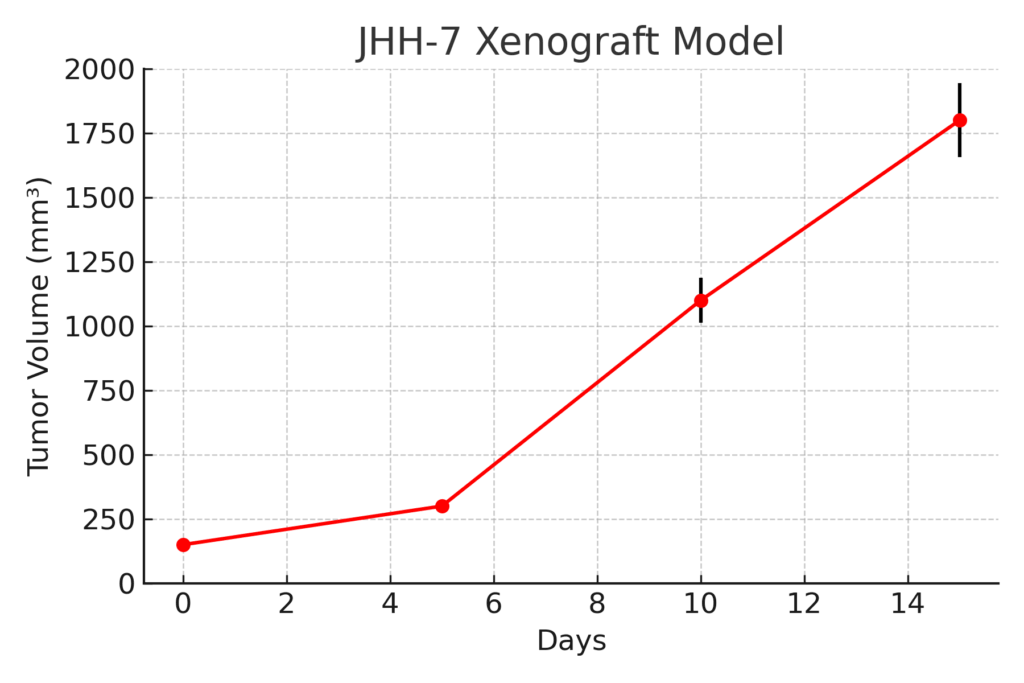
The JHH-7 xenograft model is typically developed by subcutaneous injection of cultured cells into immunodeficient mice such as BALB/c nude or NSG strains. Tumors form reliably within 10–14 days and grow progressively, often reaching volumes of 600–900 mm³ within 5–6 weeks. The model is characterized by reproducible engraftment, high tumor viability, and consistent growth kinetics, enabling longitudinal therapeutic evaluations. Orthotopic liver implantation can also be performed to more accurately replicate hepatic vascularization, parenchymal interactions, and intrahepatic metastasis. Subcutaneous xenografts, however, remain the standard for high-throughput pharmacologic testing and biomarker analysis.
Request a Custom Quote for JHH‑7 Xenograft ModelHistopathology and Immunohistochemical Profile
Histological assessment of JHH-7 tumors reveals well-differentiated, trabecular arrangements of polygonal tumor cells with abundant eosinophilic cytoplasm and centrally located nuclei. The tumors recapitulate many features of human HCC, including bile canaliculus-like structures and cytoplasmic inclusions. Immunohistochemical staining demonstrates strong expression of albumin, HNF4A, and cytokeratins, while β-catenin may be localized to the nucleus or membrane depending on Wnt pathway activation. Ki-67 indices are moderately high, indicative of proliferative capacity, and CD34 staining reveals tumor-associated neovasculature. These attributes confirm the model’s suitability for histological validation of drug-induced tumor regression or differentiation.
Preclinical Applications and Drug Response
JHH-7 xenografts have been used to evaluate targeted therapies against canonical HCC signaling pathways, including inhibitors of PI3K/AKT, TGF-β receptor kinases, and Wnt/β-catenin modulators. The model’s wild-type TP53 status allows for exploration of p53-activating compounds and DNA-damaging agents in a genetically defined context. JHH-7 is also valuable in hepatotoxicity screening, given its expression of key metabolic enzymes and bile acid regulators. Drug responses observed in this model have informed translational strategies for well-differentiated HCC subtypes and guided development of combination regimens that enhance therapeutic durability while preserving liver function.
Request This Model
To include the JHH-7 xenograft model in your hepatocellular carcinoma research pipeline or therapeutic screening assays, please reach out to our scientific team for access to validated model protocols, engraftment strategies, and customized study support tailored to liver cancer research.
Request a Custom Quote for JHH‑7 Xenograft Model