HT55 Xenograft Model Overview
The HT55 xenograft model originates from a human colorectal adenocarcinoma cell line established from the primary tumor of a 70-year-old male patient. Known for its epithelial morphology and capacity to retain structural features of colorectal origin, HT55 is widely employed in preclinical colorectal cancer research. This model is particularly relevant for studying therapeutic resistance in microsatellite-stable colorectal tumors, and it offers a robust in vivo platform for evaluating both cytotoxic agents and molecularly targeted therapies. The HT55 xenograft’s predictable tumorigenicity, preserved histopathologic characteristics, and defined molecular profile render it a reliable and translationally relevant system for drug screening and mechanistic investigations.
Request a Custom Quote for HT55 Xenograft ModelBiological and Molecular Characteristics
HT55 cells demonstrate an epithelial growth pattern with cohesive colonies and are microsatellite stable (MSS), which distinguishes them from mismatch-repair-deficient models. The cell line carries a KRAS G12D mutation, a common oncogenic driver in colorectal carcinoma that is frequently associated with resistance to EGFR-targeted therapies. TP53 is mutated in HT55, contributing to impaired apoptotic regulation, a factor that must be considered when testing DNA-damaging or p53-reliant agents. The cells express moderate levels of carcinoembryonic antigen (CEA) and epithelial cell adhesion molecule (EpCAM), both of which are retained in vivo and serve as useful biomarkers for tumor lineage confirmation and therapeutic targeting. HT55’s stable karyotype, absence of microsatellite instability, and typical chromosomal aberrations found in colorectal cancer contribute to its utility in modeling conventional CRC phenotypes.
| Characteristic | HT55 Cell Line Profile |
|---|---|
| Tissue of Origin | Colorectal adenocarcinoma |
| KRAS Status | Mutant (G12D) |
| TP53 Status | Mutant |
| CEA Expression | Moderate |
| MSI Status | Microsatellite stable (MSS) |
| Growth Characteristics | Epithelial, adherent |
In Vivo Model Development and Tumorigenicity
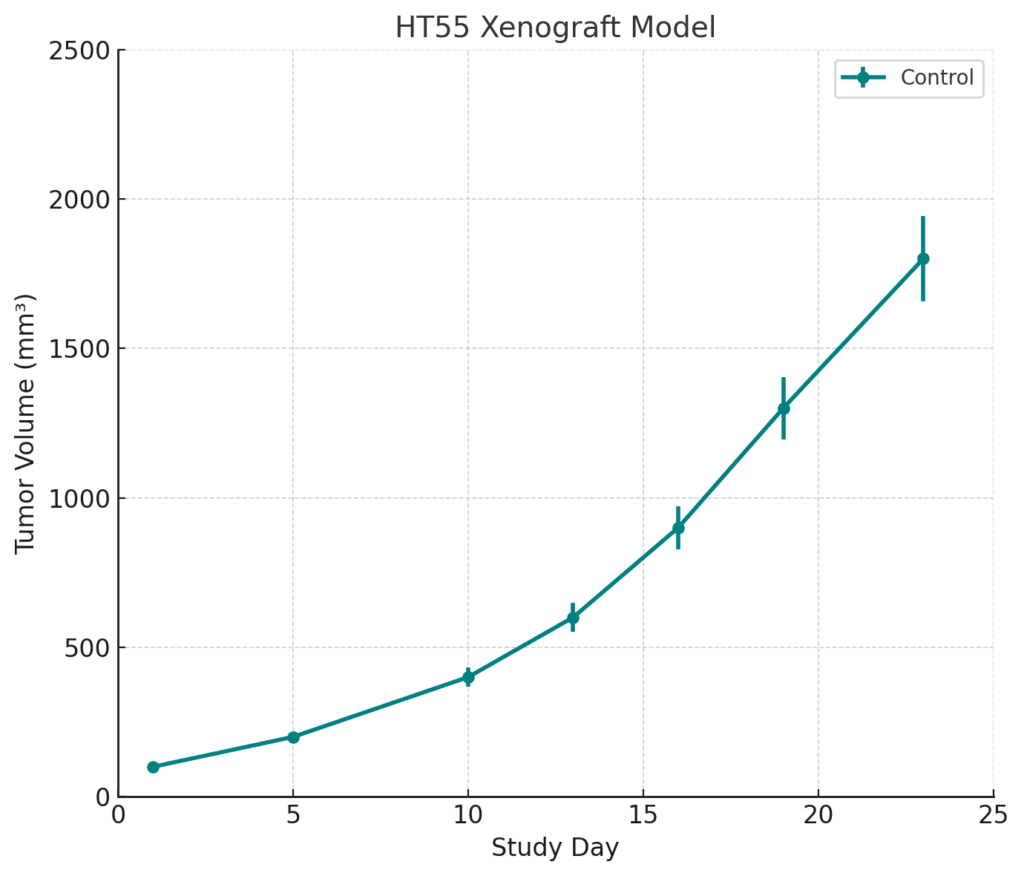
HT55 xenografts are typically established through subcutaneous injection of cultured cells into immunodeficient mouse strains such as athymic nude or NOD/SCID mice. The model exhibits consistent engraftment with initial tumor growth detectable within 10 to 14 days post-implantation. Tumor expansion follows a moderate growth trajectory, with volumes approaching 800 to 900 mm³ by day 35 in untreated control groups. This model demonstrates favorable inter-animal consistency, minimal spontaneous necrosis, and a low incidence of ulceration. Its KRAS mutation profile enables its use in studies of MAPK pathway-targeting compounds and allows for the design of combinatorial regimens that circumvent EGFR blockade resistance. The model’s tumor microenvironment supports limited stromal interaction, enabling direct assessment of tumor cell-intrinsic drug responses.
Request a Custom Quote for HT55 Xenograft ModelHistopathology and Immunohistochemical Profile
HT55 xenograft tissues maintain moderately differentiated glandular structures consistent with colorectal epithelial morphology. Histological evaluation reveals tubular and cribriform growth patterns, minimal stromal desmoplasia, and occasional mucinous differentiation. The tumors exhibit moderate mitotic activity and regions of central necrosis in later stages of growth. Immunohistochemical staining confirms strong cytoplasmic and membranous expression of CEA and EpCAM, while pan-cytokeratin and CK20 are also expressed, verifying intestinal epithelial lineage. Nuclear p53 accumulation and cytoplasmic β-catenin staining patterns reflect underlying mutational status and altered Wnt signaling activity. These features underscore the HT55 model’s value in preclinical studies aimed at biomarker analysis and mechanism-based therapy development.
Preclinical Applications and Drug Response
The HT55 xenograft model is a valuable tool for evaluating therapeutic efficacy in KRAS-mutant, MSS colorectal cancer. Its resistance to anti-EGFR therapies such as cetuximab and panitumumab mirrors clinical outcomes, making it suitable for testing next-generation inhibitors that bypass KRAS-driven resistance mechanisms. Targeted inhibitors of the MEK, ERK, and PI3K pathways have shown antitumor activity in this model, especially when used in combination to suppress compensatory signaling. The model is also useful for studying the activity of DNA-damaging agents, although responses may be attenuated due to TP53 dysfunction. HT55 xenografts are compatible with pharmacokinetic and pharmacodynamic evaluations, tissue-based biomarker studies, and mechanistic research into apoptosis resistance, oncogenic signaling, and immune evasion. Their stable growth pattern and reproducible phenotype support their use in controlled, multi-arm therapeutic evaluations.
Request This Model
For researchers developing or validating colorectal cancer therapies, the HT55 xenograft model offers a reliable and mechanistically informative platform. Connect with our scientific team to customize your study design and access this model for preclinical investigations.
Request a Custom Quote for HT55 Xenograft Model