DMS114 Xenograft Model Overview
The DMS114 xenograft model is derived from a human small cell lung carcinoma (SCLC) and provides a preclinical platform for studying high-grade neuroendocrine tumors of pulmonary origin. The DMS114 cell line was established from a lymph node metastasis of a male patient with SCLC and is notable for its relatively non-classical neuroendocrine features and slower proliferation rate compared to other SCLC models. This xenograft system is particularly valuable for evaluating therapies targeting neuroendocrine differentiation, DNA damage response, and anti-apoptotic signaling in SCLC. When implanted subcutaneously into immunodeficient mice, DMS114 cells produce compact tumors with neuroendocrine histological characteristics and reproducible growth kinetics. The model’s unique molecular profile supports its use in therapeutic research that extends beyond conventional platinum-etoposide regimens, including targeted and combination-based strategies.
Request a Custom Quote for DMS114 Xenograft ModelBiological and Molecular Characteristics
DMS114 cells exhibit epithelial morphology with partial neuroendocrine marker expression, diverging from the highly proliferative and poorly differentiated phenotype typical of classical SCLC. The cell line is wild-type for KRAS and EGFR and does not harbor MYC amplification, but it demonstrates inactivation of TP53 and RB1, hallmark alterations associated with SCLC pathogenesis. It expresses variable levels of neuroendocrine markers such as synaptophysin and neuron-specific enolase (NSE), but lacks strong chromogranin A expression, making it a representative of the “variant” SCLC subtype. The model also shows moderate expression of BCL-2, survivin, and other anti-apoptotic proteins, making it appropriate for studies targeting apoptotic resistance mechanisms. PD-L1 expression is generally low, but the model retains functional MHC class I, permitting exploration of immune sensitization strategies.
| Characteristic | Description |
|---|---|
| Tissue Origin | Human small cell lung carcinoma (lymph node metastasis) |
| Key Genetic Features | TP53 and RB1 inactivation, KRAS/EGFR wild-type |
| Cell Morphology | Epithelial, adherent, partial neuroendocrine differentiation |
| Neuroendocrine Markers | Synaptophysin+, NSE+, Chromogranin A (weak/negative) |
| Apoptotic Profile | Moderate BCL-2, survivin expression |
In Vivo Model Development and Tumorigenicity
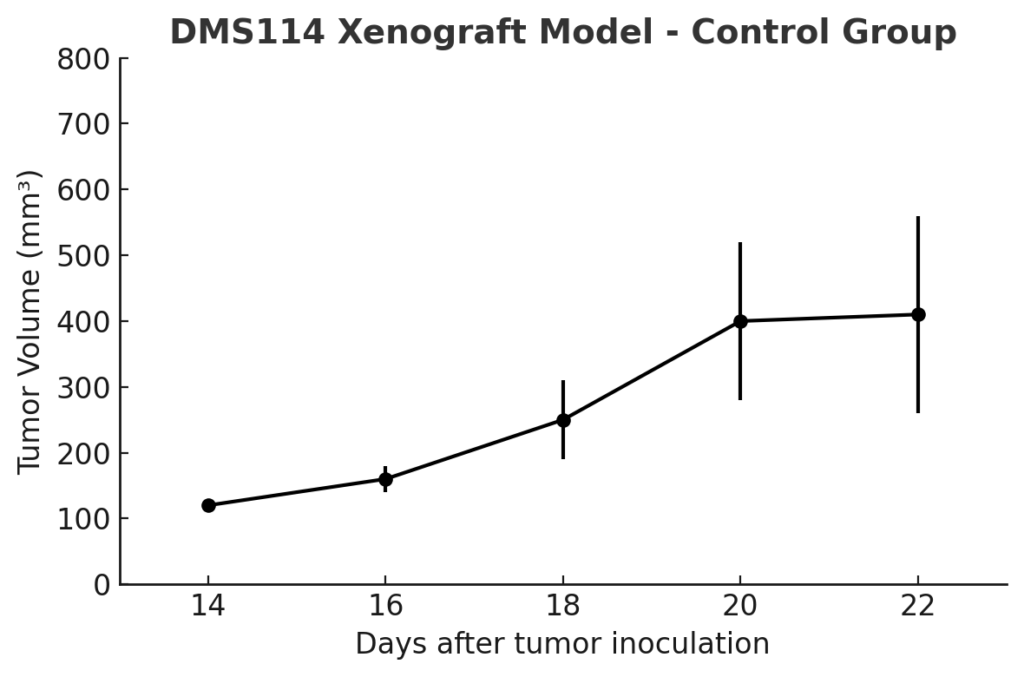
The DMS114 xenograft model is established by subcutaneous injection into immunodeficient mouse strains, including nude or NOD/SCID mice. Compared to other SCLC models, tumor onset is moderate, with growth detectable within 14–18 days post-injection and tumor volumes of 300–500 mm³ typically achieved by week six. The tumors exhibit cohesive architecture and relatively low necrosis, allowing for detailed histological and pharmacodynamic analysis. Given its reduced proliferative index and neuroendocrine variability, the model is suited for evaluating compounds that rely on tumor cell differentiation or modulation of apoptotic thresholds rather than indiscriminate cytotoxicity. It is compatible with repeat dosing and long-term study timelines, supporting both monotherapy and combination treatment designs.
Request a Custom Quote for DMS114 Xenograft ModelHistopathology and Immunohistochemical Profile
Histological examination of DMS114 xenografts reveals densely cellular tumors with uniform nuclear morphology and scant cytoplasm, consistent with neuroendocrine carcinoma. Hematoxylin and eosin staining highlights the presence of fine nuclear chromatin, rosette-like structures, and minimal mitotic activity relative to other SCLC lines. The Ki-67 proliferation index generally ranges from 30% to 50%, reflecting a moderate growth rate. Immunohistochemistry confirms synaptophysin and NSE expression, while chromogranin A is weak or absent, consistent with a less differentiated neuroendocrine phenotype. E-cadherin and cytokeratin staining validate the epithelial origin, and p53 protein accumulation is consistent with mutational inactivation. PD-L1 expression is low, but sufficient to investigate enhancement strategies through immune checkpoint modulation or cytokine priming.
Preclinical Applications and Drug Response
The DMS114 xenograft model is a versatile tool for preclinical studies focusing on variant SCLC biology. Its distinct neuroendocrine profile and moderate growth kinetics make it ideal for evaluating therapeutic agents targeting DNA damage response pathways, including PARP inhibitors and ATR/CHK1 inhibitors. Additionally, the model supports research into apoptosis-sensitizing agents such as BCL-2 antagonists, survivin inhibitors, and agents that restore p53 pathway activity. Its modest PD-L1 expression and intact antigen presentation machinery allow for investigation into immune-modulating therapies, including combinations with checkpoint inhibitors and cytokine-based agents. The DMS114 model is also suited for evaluating differentiation-inducing compounds and experimental regimens aiming to convert resistant neuroendocrine tumors into immunologically visible or drug-sensitive states.
Request This Model
To request the DMS114 xenograft model for your preclinical studies, please use the form below. A customized quote and additional model specifications will be provided upon inquiry.
Request a Custom Quote for DMS114 Xenograft Model