COLO 201 Xenograft Model Overview
The COLO 201 xenograft model is derived from a human colorectal adenocarcinoma cell line established from the ascitic fluid of a 70-year-old male patient with metastatic colon cancer. This model exhibits characteristics of well-differentiated colorectal carcinoma and is commonly used in translational oncology studies focused on epithelial biology, chemotherapy response, and drug resistance in microsatellite-stable tumors. COLO 201 cells form highly organized epithelial layers and retain classical glandular features in vivo, making the xenograft particularly useful for evaluating epithelial-directed therapies, Wnt signaling inhibitors, and targeted agents for KRAS wild-type colorectal cancer. The consistent tumor take rate and reproducible growth kinetics enhance its utility in long-term pharmacodynamic and efficacy investigations.
Request a Custom Quote for COLO 201 Xenograft ModelBiological and Molecular Characteristics
COLO 201 cells display robust epithelial morphology, forming cohesive colonies with strong cell–cell adhesion. The line is microsatellite stable (MSS) and carries wild-type KRAS and BRAF, supporting its responsiveness to EGFR-targeted therapeutics. TP53 is mutated, which can affect DNA damage repair pathways and modulate apoptotic signaling. The cell line expresses high levels of E-cadherin, cytokeratin 20 (CK20), and carcinoembryonic antigen (CEA), indicating its preserved colorectal epithelial identity. Wnt/β-catenin signaling is active, with predominant membrane-bound and cytoplasmic localization of β-catenin. These molecular traits position COLO 201 as an appropriate model for testing EGFR inhibitors, differentiating agents, and transcriptional regulators within the context of MSS colorectal cancer.
| Characteristic | COLO 201 Cell Line Profile |
|---|---|
| Tissue of Origin | Colorectal adenocarcinoma (ascites) |
| KRAS/BRAF Status | Wild-type |
| TP53 Status | Mutated |
| MSI Status | Microsatellite stable (MSS) |
| Differentiation Markers | CK20, CEA, E-cadherin |
| Wnt Signaling | Active (non-nuclear β-catenin) |
In Vivo Model Development and Tumorigenicity
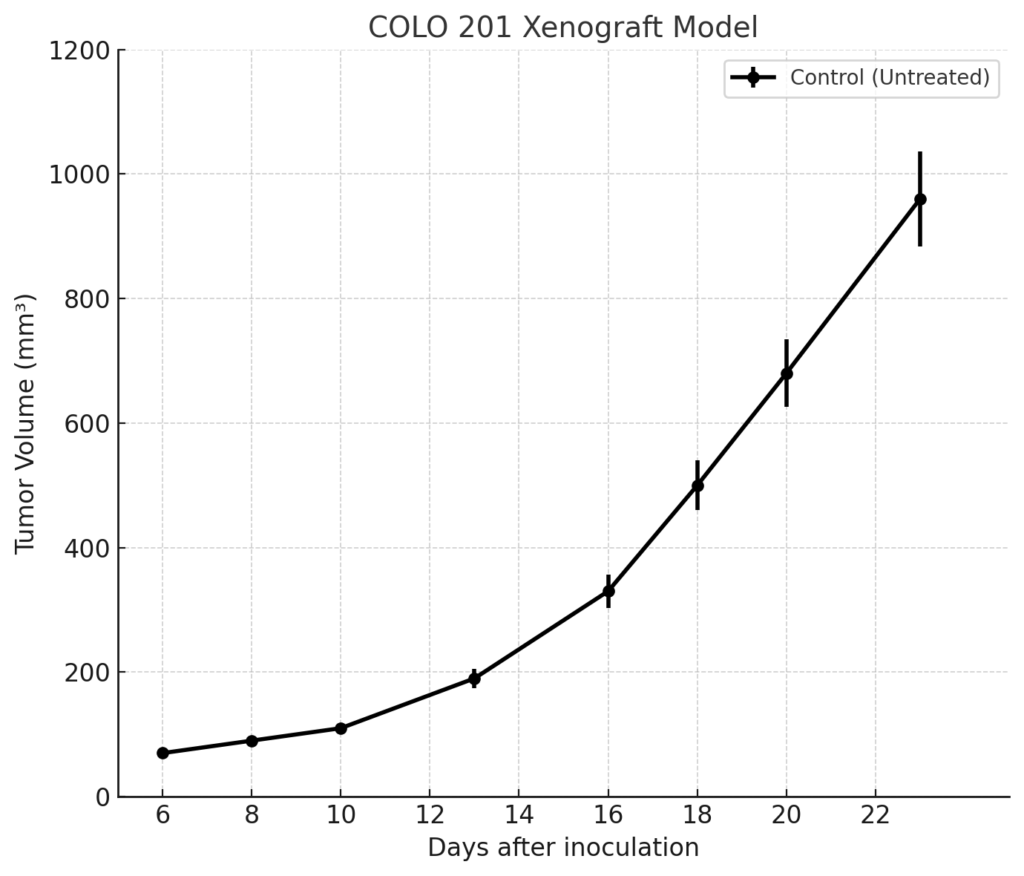
COLO 201 xenografts are typically generated by subcutaneous injection of cultured cells into immunocompromised murine models such as athymic nude or NOD/SCID mice. Tumor engraftment is rapid and reliable, with detectable growth typically observed within 7 to 10 days. Tumor volumes generally reach 700 to 900 mm³ by day 28–35 post-implantation, providing a stable window for evaluating antitumor efficacy and pharmacodynamic endpoints. The model’s well-differentiated phenotype supports longitudinal studies and extended treatment regimens without extensive necrosis or tumor ulceration. Its KRAS/BRAF wild-type status and stable genomic background allow for clean evaluation of EGFR-directed and Wnt-pathway-targeting therapeutics, as well as combination approaches that address feedback resistance mechanisms.
Request a Custom Quote for COLO 201 Xenograft ModelHistopathology and Immunohistochemical Profile
Histologically, COLO 201 xenograft tumors exhibit glandular architecture with luminal spaces and well-polarized columnar epithelial cells. Hematoxylin and eosin (H&E) staining reveals low mitotic activity and moderate stromal infiltration, contributing to a stable and uniform histologic presentation. Immunohistochemical analysis confirms strong membrane expression of E-cadherin and β-catenin, as well as positive staining for CK20 and CEA, indicating preserved epithelial differentiation and colorectal origin. Nuclear p53 accumulation is commonly observed due to the underlying TP53 mutation. Minimal necrosis and well-maintained tumor architecture further support the model’s use in studies requiring consistent histological features over extended time courses.
Preclinical Applications and Drug Response
The COLO 201 xenograft model is well-suited for evaluating EGFR-targeted therapies such as cetuximab and panitumumab, due to its KRAS/BRAF wild-type background. It also provides a reliable system for testing Wnt/β-catenin inhibitors, transcriptional co-regulator antagonists, and differentiation-inducing agents. The model is moderately sensitive to DNA-damaging chemotherapeutics, although TP53 mutation may confer reduced apoptotic responsiveness. Its epithelial integrity and consistent tumor structure allow for comprehensive PK/PD profiling and tissue biomarker analysis. COLO 201 is also used to explore drug resistance mechanisms that arise in the absence of major oncogenic drivers, offering a tractable platform for identifying resistance-modifying genes and combination strategies in MSS colorectal cancer.
Request This Model
To integrate the COLO 201 xenograft model into your colorectal cancer research program, reach out to our team to discuss model access, custom in vivo studies, and therapeutic evaluation strategies.
Request a Custom Quote for COLO 201 Xenograft Model