HCC1428 Xenograft Model Overview
The HCC1428 xenograft model is derived from a primary ductal carcinoma of the breast and belongs to the luminal A molecular subtype. Originating from a patient with estrogen receptor-positive, HER2-negative disease, the HCC1428 cell line exhibits strong hormone sensitivity and retains functional ER signaling pathways. It is widely used in preclinical studies of hormone-dependent breast cancer, particularly in investigations of endocrine therapy responsiveness, resistance evolution, and estrogen signaling crosstalk with other proliferative pathways.
This model is notable for its relatively slow in vivo growth and low baseline proliferation rate, features that closely resemble indolent luminal A tumors in clinical settings. HCC1428 is therefore well suited for long-term therapeutic experiments, minimal residual disease modeling, and evaluation of agents that act over extended durations, such as selective estrogen receptor degraders (SERDs), aromatase inhibitors, and CDK4/6 inhibitors.
Request a Custom Quote for HCC1428 Xenograft ModelBiological and Molecular Characteristics
HCC1428 cells exhibit a canonical luminal A profile, defined by strong expression of estrogen receptor alpha (ERα), partial expression of progesterone receptor (PR), and absence of HER2 amplification. These cells are hormone-responsive and proliferate in an estrogen-dependent manner. They also demonstrate low expression of basal cytokeratins, low Ki-67 levels, and an intact BRCA1/2 background.
Mutational analysis reveals a wild-type TP53 genotype and low mutational burden overall, consistent with a genetically stable phenotype. While the PI3K pathway is active at baseline, it does not harbor PIK3CA or PTEN mutations, distinguishing it from more aggressive luminal subtypes. The low proliferative index and intact apoptotic machinery make this model ideal for dissecting early resistance to anti-estrogen therapies.
| Characteristic | HCC1428 Profile |
|---|---|
| Tumor Type | Human breast ductal carcinoma |
| Receptor Status | ER+, PR±, HER2– |
| Molecular Subtype | Luminal A |
| TP53 Status | Wild-type |
| BRCA1/2 Status | Wild-type |
| Proliferation Rate | Low (Ki-67 < 20%) |
| Hormone Responsiveness | Estrogen-dependent; responsive to tamoxifen, fulvestrant |
| PI3K/AKT Pathway | Baseline activation; no hotspot mutations |
| Cytokeratin Expression | Low basal cytokeratin expression |
These features make HCC1428 a useful platform for testing hormone-dependent tumor biology and developing strategies to prevent or reverse endocrine resistance.
In Vivo Model Development and Tumorigenicity
HCC1428 xenografts are typically established by subcutaneous injection of 5 × 10⁶ cells into estrogen-supplemented immunodeficient female mice, such as ovariectomized athymic nude or NOD/SCID strains. Estrogen supplementation, usually via subcutaneous 17β-estradiol pellets or continuous-release pumps, is required for optimal tumor take and sustained growth. Without hormonal support, tumor engraftment rates decline significantly due to the model’s strict estrogen dependence.
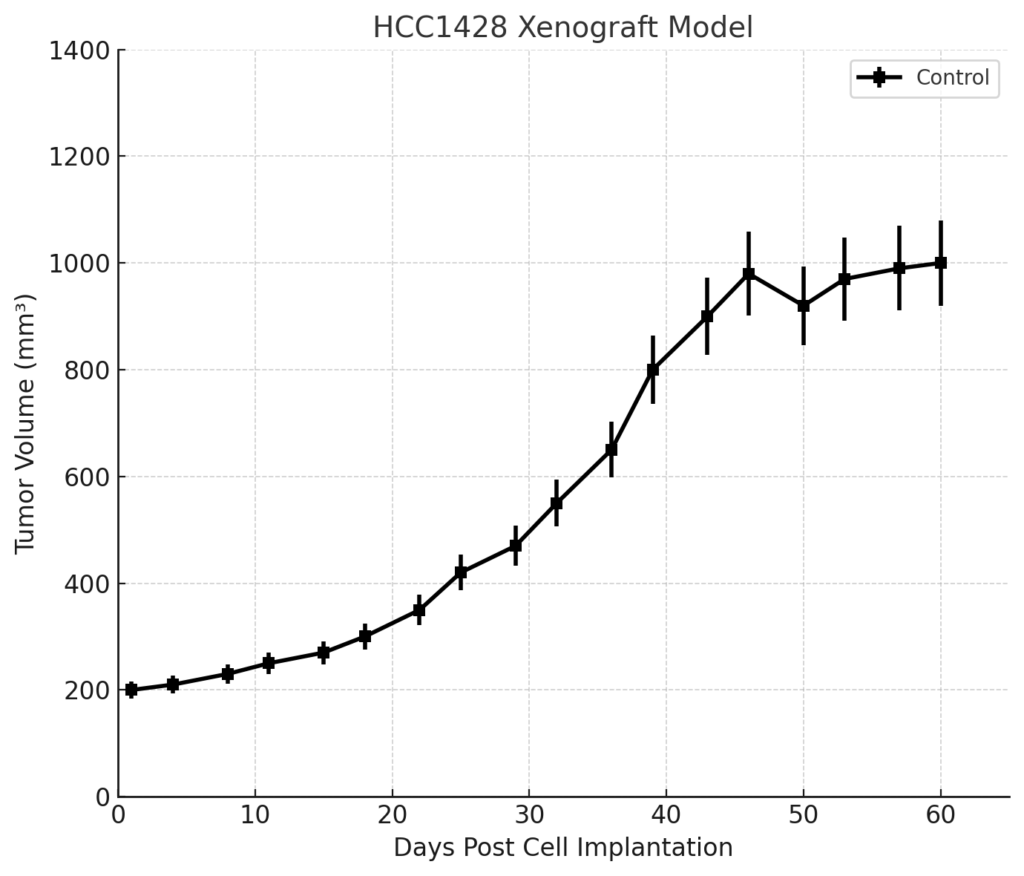
Tumors form slowly compared to basal-like or HER2-enriched xenografts, often taking 2–3 weeks to become palpable and 6–8 weeks to reach endpoint volumes of 1,000–1,200 mm³. This slow growth profile makes the model particularly useful for evaluating chronic exposure to endocrine therapies or investigating early molecular changes preceding resistance.
The model is compatible with orthotopic implantation into the mammary fat pad and can be transduced with luciferase or GFP reporters for non-invasive imaging studies.
Request a Custom Quote for HCC1428 Xenograft ModelHistopathology and Immunohistochemical Profile
HCC1428 xenografts present histologically as well-differentiated ductal carcinomas with glandular morphology, low mitotic index, and uniform nuclear features. Necrosis is minimal, and tumor architecture is typically organized in nests or tubules surrounded by fibrous stroma. These tumors often resemble low-grade ER+ clinical specimens.
Immunohistochemical analysis confirms strong nuclear ER expression in the majority of tumor cells, with partial PR positivity. HER2 is absent by both IHC and FISH. Ki-67 staining levels remain low (<20%), aligning with the luminal A phenotype. Apoptosis markers such as BAX and cleaved caspase-3 are detectable at baseline and can be modulated by treatment with SERMs or SERDs. Phosphorylated AKT and S6 may be modestly expressed, depending on in vivo conditions and treatment history.
The IHC profile validates the hormone-sensitive nature of this model and supports its utility in studies of ER-driven tumorigenesis and therapy adaptation.
Preclinical Applications and Drug Response
HCC1428 xenografts are a gold-standard model for evaluating ER-targeted therapies, including tamoxifen, fulvestrant, and next-generation SERDs. The model has also been used to assess aromatase inhibitors in combination with exogenous hormone withdrawal. It serves as an important comparator for testing resistance-inducing conditions such as long-term estrogen deprivation or chronic drug exposure.
Because of its relatively quiescent cell cycle status, HCC1428 is valuable in assessing CDK4/6 inhibitors (e.g., palbociclib, abemaciclib) and determining their effects on tumor dormancy and cell cycle re-entry. The model is also useful for exploring epigenetic modifiers that may prime or restore endocrine sensitivity, and for identifying biomarkers predictive of late recurrence in ER+ breast cancer.
The model’s low background mutational complexity enables clear attribution of drug effects and is ideal for mechanistic studies involving hormone signaling, apoptosis regulation, and transcriptional reprogramming during acquired resistance.
Request This Model
To incorporate the HCC1428 xenograft model into endocrine therapy studies, resistance modeling, or luminal A subtype drug development programs, use the quote request link below. Services include estrogen-supplemented tumor engraftment, pharmacodynamic endpoint analysis, and custom therapy design.
Request a Custom Quote for HCC1428 Xenograft Model