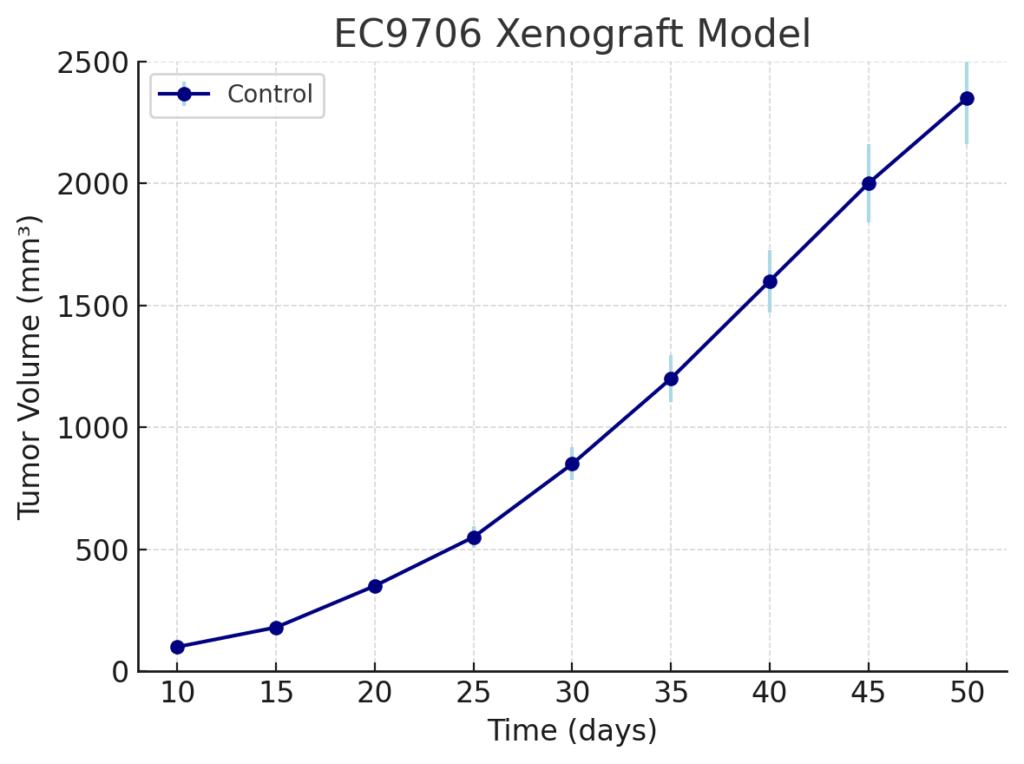
EC9706 Xenograft Model Overview
The EC9706 xenograft model originates from a human esophageal squamous cell carcinoma (ESCC) cell line, established from a poorly differentiated tumor in a 61-year-old male patient in China. It has become a widely used tool for modeling ESCC in vivo, particularly due to its robust tumorigenic capacity, aggressive growth kinetics, and relevance to high-incidence regions where squamous histology predominates. EC9706 xenografts serve as an effective preclinical platform for investigating radiation sensitization, chemotherapy response, molecular signaling in squamous epithelium, and the biological mechanisms driving treatment resistance. The model supports reproducible subcutaneous tumor formation and reflects key histopathological features of aggressive ESCC, making it essential for translational oncology studies targeting this malignancy.
Request a Custom Quote for EC9706 Xenograft ModelBiological and Molecular Characteristics
EC9706 cells exhibit classic squamous epithelial morphology, forming tightly adherent monolayers in culture. The line is characterized by TP53 mutation, frequently detected in ESCC, and alterations in pathways associated with cell cycle dysregulation and DNA repair. It exhibits wild-type KRAS and BRAF but shows high levels of epidermal growth factor receptor (EGFR) expression, contributing to its rapid proliferation and sensitivity to EGFR inhibition. Additionally, EC9706 demonstrates nuclear expression of SOX2, a transcription factor associated with stemness and squamous identity. High expression of squamous markers, including cytokeratin 5/6 (CK5/6), p63, and involucrin, reinforces its classification as a representative model of ESCC. These features enable targeted studies of squamous-specific oncogenic pathways and therapeutic vulnerabilities in esophageal carcinoma.
| Characteristic | EC9706 Cell Line Profile |
|---|---|
| Tissue of Origin | Esophageal squamous cell carcinoma (ESCC) |
| KRAS/BRAF Status | Wild-type |
| TP53 Status | Mutated |
| EGFR Expression | High |
| Squamous Markers | CK5/6, p63, involucrin |
| Stemness Markers | SOX2-positive |
In Vivo Model Development and Tumorigenicity
EC9706 xenografts are developed through subcutaneous injection of cultured tumor cells into immunodeficient mice such as BALB/c nude or NOD/SCID strains. Tumor engraftment occurs reliably within 7 to 10 days post-implantation, with tumors reaching volumes of 700–900 mm³ in 3 to 4 weeks. This rapid tumor growth enables efficient study timelines, while the model’s consistent engraftment supports robust comparisons across treatment groups. EC9706 tumors are known for their high mitotic index and aggressive local invasion, facilitating studies on proliferation, metastatic potential, and treatment efficacy. The model is also amenable to orthotopic implantation into the esophagus for studies requiring anatomical relevance and local tumor–host interactions.
Request a Custom Quote for EC9706 Xenograft ModelHistopathology and Immunohistochemical Profile
Histologically, EC9706 xenografts exhibit poorly differentiated squamous cell carcinoma morphology with solid nests, intercellular bridges, and occasional keratin pearl formation. Hematoxylin and eosin (H&E) staining reveals hyperchromatic nuclei, irregular nuclear contours, and high mitotic activity. Immunohistochemical analysis confirms strong and diffuse expression of squamous markers CK5/6 and p63, with EGFR localized at the cell membrane. SOX2 expression is nuclear and prominent, reinforcing the tumor’s squamous and stem-like identity. Involucrin is present in differentiated zones, while mutant p53 protein accumulates in tumor nuclei. This profile underscores the model’s histologic and molecular fidelity to human ESCC and supports its use in therapy evaluation and biomarker discovery.
Preclinical Applications and Drug Response
The EC9706 xenograft model is extensively used in preclinical studies investigating ESCC-specific therapies, especially those targeting EGFR signaling, TP53-related DNA damage responses, and squamous differentiation. The model is responsive to EGFR inhibitors such as gefitinib and erlotinib, although resistance mechanisms frequently develop, making it suitable for combination therapy studies and resistance reversal strategies. Due to its TP53 mutation, EC9706 is also ideal for testing radiation and cisplatin-based regimens in the context of impaired apoptosis. In addition, the model supports evaluation of SOX2-targeted strategies, Wnt pathway inhibitors, and immunomodulatory agents. Its rapid tumor growth, reproducibility, and squamous specificity make EC9706 a cornerstone in esophageal cancer drug development pipelines.
Request This Model
To incorporate the EC9706 xenograft model into your esophageal cancer research program or preclinical development strategy, contact our scientific team to request detailed specifications, discuss customized study options, and gain access to this validated and clinically relevant ESCC model.
Request a Custom Quote for EC9706 Xenograft Model