5637 Xenograft Model Overview
The 5637 xenograft model is derived from a grade II urinary bladder carcinoma and serves as a robust system for modeling intermediate-to-high-grade non-muscle-invasive bladder cancer (NMIBC). The parental 5637 cell line was established from a primary tumor of a 68-year-old male patient and is distinguished by its partial differentiation, high proliferative index, and moderate invasiveness. The model is frequently used in preclinical research to evaluate chemotherapeutic responses, immune modulation, and cell cycle dysregulation in bladder epithelium that has not yet transitioned into a fully muscle-invasive phenotype.
The 5637 xenograft offers consistent tumor formation, reliable growth kinetics, and preserved epithelial features. It occupies a niche between well-differentiated luminal models (e.g., RT4, RT112) and aggressive mesenchymal subtypes (e.g., UM-UC-3, T24). This makes it particularly suitable for investigating therapeutic strategies in transitional epithelial states, including DNA-damaging agents, epigenetic drugs, and immune-activating regimens. It is also used to study p53-associated resistance mechanisms and stress-induced apoptosis in intermediate-grade bladder carcinoma.
Request a Custom Quote for 5637 Xenograft ModelBiological and Molecular Characteristics
The 5637 cell line exhibits a mutant TP53 gene and corresponding non-functional p53 protein accumulation, leading to impaired DNA damage response and apoptotic resistance. Although the cells display high levels of E-cadherin and cytokeratin 18, indicative of epithelial identity, they also demonstrate mesenchymal transition features under stress conditions, including upregulation of vimentin and fibronectin. This hybrid phenotype allows for investigations into epithelial-mesenchymal plasticity and drug-induced dedifferentiation.
The 5637 line expresses moderate levels of EGFR, CD44, and MMP-9, which contribute to proliferation, adhesion, and matrix remodeling. Notably, it lacks FGFR3 mutations and is wild-type for RB1, allowing intact G1/S checkpoint regulation in the absence of external stressors. The PI3K/AKT and MAPK/ERK pathways are both constitutively active at basal levels, with further upregulation under hypoxia or drug exposure.
The table below summarizes the major biological and molecular features of the 5637 cell line:
| Characteristic | 5637 Profile |
|---|---|
| Origin | Human bladder carcinoma, grade II |
| TP53 Status | Mutated (non-functional p53 accumulation) |
| RB1 Status | Wild-type |
| EGFR Expression | Moderate |
| FGFR3 Status | Wild-type |
| Differentiation Markers | CK18+, E-cadherin+ |
| EMT Profile | Partial; inducible under stress |
| MMP and Invasion Markers | MMP-9+, CD44+ |
| Growth Factor Signaling | PI3K/AKT and ERK1/2 activated |
| Immunogenicity Markers | HLA class I+, PD-L1 inducible |
These characteristics position 5637 as a versatile model for studying molecularly defined transitions between epithelial stability and mesenchymal adaptation, especially in the presence of drug-induced stress or immune checkpoint activity.
In Vivo Model Development and Tumorigenicity
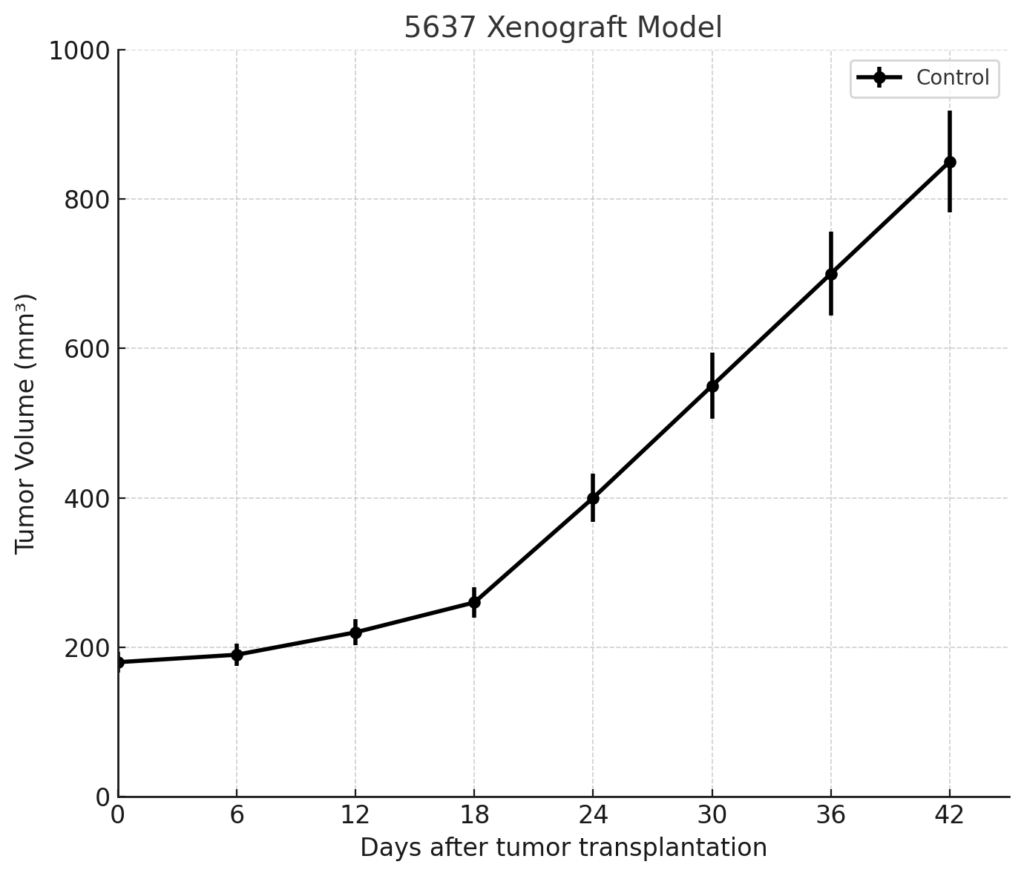
The 5637 xenograft model forms tumors with high reliability in immunodeficient mice. Subcutaneous injection of 5 × 10^6 to 1 × 10^7 cells suspended in Matrigel yields tumor take rates of 85–95% in athymic nude or NOD/SCID mice. Tumors are typically palpable within 10–14 days post-injection and demonstrate steady volumetric expansion over 3–4 weeks, with endpoint volumes (1,200–1,500 mm³) generally achieved by day 30–35.
Tumors grow as compact, non-metastatic nodules, with minimal evidence of stromal invasion or distant dissemination under standard subcutaneous protocols. However, orthotopic implantation via bladder wall injection has been used to examine local progression, stromal reorganization, and epithelial barrier disruption. These orthotopic models recapitulate aspects of the tumor microenvironment relevant to early-stage bladder carcinogenesis.
The 5637 model supports longitudinal tumor volume measurement, pharmacodynamic endpoint analysis, and fluorescence- or luciferase-based imaging when appropriately labeled. Its moderate growth rate makes it ideal for treatment windows of two to four weeks, accommodating both monotherapy and combination regimen testing.
Request a Custom Quote for 5637 Xenograft ModelHistopathology and Immunohistochemical Profile
Histologically, 5637 xenograft tumors appear as moderately differentiated epithelial neoplasms, composed of polygonal to round cells arranged in nests and sheets. The nuclei are large and irregular with visible nucleoli, and the mitotic rate is elevated, though necrosis is generally focal and limited. Tumor architecture is cohesive, with clear cellular borders and infrequent stromal infiltration.
Immunohistochemically, xenograft tumors stain strongly for E-cadherin and CK18, confirming their epithelial origin. Ki-67 staining indicates a proliferation index between 50–65%, while EGFR shows moderate membranous staining. Phospho-AKT and phospho-ERK1/2 are diffusely cytoplasmic, consistent with active oncogenic signaling. Under hypoxic or treatment conditions, vimentin expression increases along tumor margins, particularly in areas of higher mitotic stress, suggesting EMT plasticity.
Additional staining for MMP-9 and CD44 is positive, particularly in invasive fronts, although the model remains non-metastatic in standard murine hosts. Inducible PD-L1 expression has been observed under IFN-γ stimulation, suggesting relevance in immune checkpoint studies using humanized systems or co-engrafted PBMCs.
Preclinical Applications and Drug Response
The 5637 xenograft model is widely used for evaluating drugs that target DNA replication stress, checkpoint kinases (CHK1/2, WEE1), and apoptosis-inducing agents in p53-deficient backgrounds. The model demonstrates moderate resistance to cisplatin and doxorubicin, which can be partially reversed by HDAC inhibitors or BCL-2 family antagonists, making it a valuable system for combination therapy screening.
Due to its constitutively active AKT and ERK pathways, the model responds to PI3K/AKT and MEK inhibitors, especially under dual blockade conditions. It is also suited for studying epigenetic modulators (e.g., DNMT inhibitors, BET bromodomain inhibitors) aimed at restoring p53-independent apoptotic control or suppressing EMT progression.
Recent applications include testing of immunomodulatory agents under humanized conditions, particularly drugs affecting MHC-I regulation, PD-L1 expression, or tumor antigen presentation. Its moderate growth rate allows for longitudinal monitoring of treatment response, biomarker dynamics, and resistance development.
Request This Model
To request the 5637 xenograft model or discuss experimental configurations including checkpoint inhibitor testing, drug resistance profiling, or EMT suppression studies, please use the form below. We will support study design, technical parameters, and timeline planning.
Request a Custom Quote for 5637 Xenograft Model